Intranasal Glucagon for the treatment of hypoglycemia - abstracted references:
(1988). "Intranasal glucagon for hypoglycaemia
[letter]." Lancet 2(8604): 216-7.
Aranishi, T., Y. Nagai, et al.
(2020). "Usability of Nasal Glucagon Device: Partially Randomized
Caregiver and Third-Party User Experience Trial with Simulated
Administration at a Japanese Site." Diabetes Ther 11(1): 197-211.
Boido, A., V. Ceriani, et al. (2014). "Glucagon for hypoglycemic
episodes in insulin-treated diabetic patients: a systematic review and
meta-analysis with a comparison of glucagon with dextrose and of
different glucagon formulations." Acta Diabetol.
AIMS: Glucagon is used as an emergency drug in hypoglycemia, mainly when
the patient is unconscious. A few studies report on ineffectiveness of
glucagon in relieving hypoglycemia. The present systematic review and
meta-analysis evaluate the effectiveness of glucagon alone and in
comparison with dextrose and the effectiveness of intranasal glucagon in
comparison with injected glucagon. METHODS: Studies were grouped into
three groups: (1) reports on glucagon ineffectiveness; (2) comparison of
glucagon and dextrose; (3) comparison of intranasal glucagon and
injected glucagon. In groups 2 and 3, only controlled studies were
included in the analysis, whether randomized or non-randomized studies.
Appropriate methodology (PRISMA statement) was adhered to, and
publication bias was formally assessed. Sixteen studies, published in
any language as full papers, were analysed to identify predictors of
ineffectiveness, and they were included in a meta-analysis (random
effects model) to study the effect of different strategies. Intervention
effect (number of failures) was expressed as odds ratio (OR), with 95 %
confidence intervals. RESULTS: Failure rate ranged from 0.0 to 2.31 %,
to 7.6 %, to 14.4 %, and to 59 %. Comparing glucagon and dextrose, the
OR was 0.53 (0.20-1.42); comparing intranasal and intramuscular
glucagon, the OR was 1.40 (0.18-10.93). Heterogeneity was low and not
statistically significant. Publication bias was absent. CONCLUSIONS:
These data indicate that ineffectiveness of glucagon is unfrequent, not
different from dextrose; in addition, intranasal and injected glucagon
are similarly effective. In the case of failure, a second dose can be
administered.
Borden, T. J., D. E. Baker, et
al. (2020). "Newly Approved Novel Dosage Forms of Glucagon for
Management of Severe Hypoglycemia." Sr Care Pharm 35(4): 176-181.
Carstens, S. and I. Andersen (1994).
"[Intranasal glucagon in the treatment of hypoglycemia. A therapeutic
possibility in the future]." Ugeskr Laeger 156(30): 4339-42.
Nearly 10% of IDDM patients receiving conventional insulin treatment and about three times as many in intensive insulin therapy yearly experience severe hypoglycaemia (requiring external assistance) The conventional treatment of severe hypoglycaemia is glucagon given intramuscularly by a relative or glucose administered intravenously by a physician. These are however not optimal treatments. Obtaining intravenous access requires a medical doctor and glucagon injection is not always properly done by family members. Glucagon administered intranasally has been proven to raise blood glucose levels in volunteers. The effect of intranasal glucagon on blood glucose is similar to that seen after intramuscular administration for the first 15 minutes following administration. However, intranasal glucagon seems more physiological in that is stabilizes blood glucose concentrations at nearfasting levels, whereas glucagon given intramuscularly tends to give hyperglycaemia. Intranasal glucagon is easy to administer, and can thus prevent serious hypoglycaemic crises and thereby make diabetics and their families more secure.
Freychet, L., S. W. Rizkalla, et al. (1988).
"Effect of intranasal glucagon on blood glucose levels in healthy
subjects and hypoglycaemic patients with insulin-dependent diabetes."
Lancet 1(8599): 1364-6.
Glucagon in solution with a surfactant (deoxycholic acid 1% w/v) was administered by intranasal spray to 6 healthy fasting subjects and 6 insulin-dependent diabetics with insulin-induced hypoglycaemia. In the normal subjects, intranasal glucagon increased plasma glucose levels, with a dose-response effect. In the diabetic patients, plasma glucose levels showed a mean increase of 100% above nadir values in approximately 26 min in response to 7.5 mg intranasal glucagon; hypoglycaemic symptoms were relieved within about 7 min. These results suggest that intranasal glucagon is effective and may represent an alternative to parenteral glucagon or glucose or to oral sugar as the first-line treatment of hypoglycaemic episodes in insulin-dependent diabetics.
Hvidberg, A., R. Djurup, et al. (1994). "Glucose
recovery after intranasal glucagon during hypoglycaemia in man." Eur
J Clin Pharmacol 46(1): 15-7.
We compared the hyperglycaemic effect of intranasal and intramuscular (i.m.) administration of glucagon after insulin-induced hypoglycaemia. Twelve healthy subjects were examined twice, receiving on both occasions an intravenous insulin bolus. Somatostatin and propranolol were administered to block endogenous glucose counterregulation, and glucose turnover was estimated by a 3-[3H]-glucose infusion. When hypoglycaemia was reached, the subjects received either i.m. glucagon of pancreatic extraction (1 mg) or intranasal genetically engineered glucagon (2 mg). The incremental values for plasma glucose concentrations 15 min after intranasal and i.m. administration of glucagon differed marginally. However, after 5 min the glucose appearance rate, as well as the incremental values for plasma glucose, were significantly higher for the i.m. glucagon treatment. The mean time taken for incremental plasma glucose to exceed 3 mmol.l-1 was significantly shorter for i.m. glucagon. The mean plasma glucagon level increased faster after i.m. glucagon than after intranasal glucagon, and the levels remained higher throughout the study period. We conclude that glucose recovery was significantly better after i.m. administration of glucagon than after intranasal administration. However, the differences between the incremental plasma glucose and the time for incremental plasma glucose to exceed 3 mmol.l-1 were not considered of major clinical importance.
Lowe, R. N. and J. M. Trujillo
(2020). "Intranasal Glucagon: A New Way to Treat Hypoglycemic
Emergencies." Ann Pharmacother 54(8): 780-787.
Objective: To review the safety, efficacy, and administration of intranasal (IN) glucagon for the management of hypoglycemia. Data Source: A literature search of PubMed/MEDLINE (1995 to November 2019) using the terms intranasal glucagon, nasal glucagon, glucagon, hypoglycemia treatment, and hypoglycemia management was completed. Study Selection and Data Extraction: English-language studies evaluating IN glucagon were evaluated. Data Synthesis: IN glucagon is a newly approved product for the treatment of hypoglycemia in patients with diabetes, 4 years and older. Administered as a 3-mg dose, it was shown to be noninferior to intramuscular (IM) glucagon. In comparison trials, more than 98% of hypoglycemic events were treated successfully with IN glucagon in both pediatric and adult patients. In simulated and real-world studies, IN glucagon was administered in less than a minute for the majority of scenarios. IM glucagon took longer to administer, ranging from 1 to 4 minutes, and often, patients did not receive the intended full dose. Nausea and vomiting, known adverse events for glucagon, as well as local adverse events were most commonly reported with IN glucagon. Relevance to Patient Care and Clinical Practice: IN glucagon is safe, effective, easy to use, and does not require reconstitution prior to use, which can lead to faster delivery in a severe hypoglycemic event. It does not require age- or weight-based dosing. This delivery method offers an option for someone who fears needles or is uncomfortable with injections. Conclusion: IN glucagon is a safe, effective, easy to use, needle-free treatment option for severe hypoglycemia.
Matsuhisa, M., Y. Takita, et al.
(2020). "Nasal glucagon as a viable alternative for treating
insulin-induced hypoglycaemia in Japanese patients with type 1 or type 2
diabetes: A phase 3 randomized crossover study." Diabetes Obes Metab
22(7): 1167-1175.
AIM: To compare nasal glucagon (NG) with intramuscular glucagon (IMG) for the treatment of insulin-induced hypoglycaemia in Japanese patients with type 1 (T1DM) or type 2 diabetes mellitus (T2DM). MATERIALS AND METHODS: This phase 3, randomized, open-label, two-treatment, two-period crossover non-inferiority study enrolled Japanese adults with T1DM or T2DM on insulin therapy, with glycated haemoglobin levels </=86 mmol/mol (</=10%). After >/=8 hours of fasting, hypoglycaemia was induced with human regular insulin (intravenous infusion). Patients received NG 3 mg or IMG 1 mg approximately 5 minutes after insulin termination. The primary endpoint was the proportion of patients achieving treatment success [plasma glucose (PG) increase to >/=3.9 mmol/L (>/=70 mg/dL) or >/=1.1 mmol/L (>/=20 mg/dL) increase from the PG nadir within 30 minutes of receiving glucagon]. Non-inferiority was declared if the upper limit of the two-sided 95% confidence interval (CI) of the mean difference in the percentage of patients achieving treatment success (IMG minus NG) was <10%. RESULTS: Seventy-five patients with T1DM (n = 34) or T2DM (n = 41) were enrolled; 72 patients (50 men, 22 women) received >/=1 study drug dose (T1DM, n = 33; T2DM, n = 39). Sixty-eight patients completed the study and were evaluable. All NG- and IMG-treated patients achieved treatment success (treatment arm difference: 0%; upper limit of two-sided 95% CI 1.47%); NG met prespecified conditions defining non-inferiority versus IMG. Glucagon was rapidly absorbed after both nasal and intramuscular administration; PG profiles were similar between administration routes during the first 60 minutes post dose. Study drug-related treatment-emergent adverse events affecting >2 patients were rhinalgia, increased blood pressure, nausea, ear pain and vomiting in the NG group, and nausea and vomiting in the IMG group. CONCLUSION: Nasal glucagon was non-inferior to IMG for successful treatment of insulin-induced hypoglycaemia in Japanese patients with T1DM/T2DM, supporting use of NG as a rescue treatment for severe hypoglycaemia.
Nakazato, M. (2011). "[Development of the novel delivery system of GLP-1
administration for the treatment of diabetes mellitus]." Nihon
Rinsho 69(5): 918-922..
Glucagon-like peptide 1 (GLP-1) is a peptide produced in the
endocrine L cells of the distal intestine. GLP-1(7-36)NH2 is a major
molecular form that stimulates insulin release, reduces food intake, and
has a potential to promote beta-cell regeneration. We have developed a
device for intranasal application of GLP-1(7-36)NH2 and completed a
double-blind clinical trial of intranasal administration of
GLP-1(7-36)NH2 to 26 type II diabetic patients. Intranasal
administration of GLP-1 increased its plasma level, stimulated
postprandial insulin release, and suppressed glucagon release. Two-week
intranasal administration of GLP-1 just before meals significantly
decreased serum glycoalbumin level and significantly increased 1,5-AG
(1,5-anhydro-D-glucitol) level. Hypoglycemia was not found through this
study. Intranasal GLP-1 administration using the novel device and
medication improved glycemic control in type 2 diabetic patients without
any adverse effects.
Pacchioni, M., C. Orena, et al. (1995). "The
hypotonic effect of intranasal and intravenous glucagon in
gastrointestinal radiology." Abdom Imaging 20(1): 44-6.
In the present study, following a double-blind, double placebo protocol vs. placebo, we compared the hypotonic effect of intranasal and intravenous glucagon during a double-contrast barium meal examination of the stomach. We found a statistically significant difference between placebo and intranasal or intravenous glucagon in inducing gastric hypomotility, with no significant differences between IN and IV glucagon. The intranasal administration of glucagon has the advantage of being noninvasive and well tolerated by the patients, and might be a valuable aid in upper gastrointestinal examination as well as in CT scan or magnetic resonance imaging of the abdomen.
Pietrzak, I. and A. Szadkowska
(2020). "Nasal glucagon - a new way to treat severe hypoglycemia in
patients with diabetes." Pediatr Endocrinol Diabetes Metab:
45-57.
Hypoglycaemia is the most frequent acute complication of diabetes in patients treated with insulin. Severe hypoglycaemia can lead to life-threatening disorders. In addition, fear of hypoglycaemia remains a major obstacle to achieving therapeutic goals in diabetics, espe-cially with type 1. As such, both the prevention and treatment of hypoglycemia are so important in diabetes care. Treatment of hypoglycemia is still based on administration of glucose (oral or parenteral depending on the level of consciousness) or of glucagon injected intramuscularly or subcutaneously. In 1983, it was shown for the first time that intranasal glucagon drops increase blood glucose levels in healthy volunteers. In subsequent years, a new powder formulation of glucagon was developed, which is applied intranasally and passively absorbed through the nasal mucosa and it is not necessary to take a deep breath to take it. Intranasal glucagon is as effective as injectable glucagon and devoid of most of the technical problems associated with injectable glucagon. No serious adverse effects of the new preparation have been de-scribed so far. In December 2019 under the name Baqsimi TM (Eli Lilly, USA) has been approved by EMA for the treatment of severe hypoglycemia in patients since 4 years of age. Intranasal glucagon appears to be a breakthrough in the treatment of severe hypoglycemia in diabetic patients treated with insulin in both children and adults.
Pontiroli, A. E. (1998). "Peptide hormones:
Review of current and emerging uses by nasal delivery." Adv Drug
Deliv Rev 29(1-2): 81-87.
The family of clinically available peptide hormones (PHs) is expanding in an exponential way, and advancement of knowledge of the basic mechanisms of action of PHs has led to multiplication of the possible clinical indications of already known PHs, and appears even more promising for still unknown PHs. A common obstacle to a full routine use of PHs is represented by the fact that PHs cannot be administered by the oral route, since they undergo digestion and inactivation in the gastrointestinal tract and a significant first pass metabolism in the liver. One alternative is represented by intranasal administration of PHs. The intranasal route of administration of PHs is also very attractive because of its convenience, which should assure a good compliance by patients. Luteinizing hormone releasing hormone, the analogues, desmopressin, oxytocin and salmon calcitonin are already marketed for intranasal administration; for salmon calcitonin, studies about bioavailability have been scanty in the past, but should be re-considered in order to fully explore its clinical benefit.Intranasal peptide hormones not yet on the market are insulin, glucagon, growth hormone releasing hormone (GHRH) and GHRP, GH and somatostatin, but the scenario is likely to change in a short period of time. Hexarelin seems very effective and is at a promising stage of development; also, glucagon appears mature enough to undergo extensive clinical evaluation and possibly marketing. The concern is why other peptides have not been further evaluated, as is the case for somatostatin and its analogues.
Pontiroli, A. E., M. Alberetto, et al. (1989).
"Nasal administration of glucagon and human calcitonin to healthy
subjects: a comparison of powders and spray solutions and of different
enhancing agents." Eur J Clin Pharmacol 37(4): 427-30.
The systemic availability of glucagon and human calcitonin given intranasally to healthy volunteers as spray solutions or powders has been examined. Glucagon was absorbed only when surfactants were used, and 9-lauryl ether (as a spray) and sodium glycocholate (as spray or powder) were equally active. Calcitonin was poorly absorbed when given alone but the surfactants dihydrofusinate (as spray or powder) and glycocholate (as a spray) were equally active in promoting absorption. Thus, enhancers are required to obtain significant nasal absorption of glucagon and calcitonin and powders and spray solutions did not differ in terms of systemic availability.
Pontiroli, A. E., M. Alberetto, et al. (1983).
"Intranasal glucagon raises blood glucose concentrations in healthy
volunteers." Br Med J (Clin Res Ed) 287(6390): 462-3.
Pontiroli, A. E., M. Alberetto, et al. (1985).
"Metabolic effects of intranasally administered glucagon: comparison
with intramuscular and intravenous injection." Acta Diabetol Lat
22(2): 103-10.
Intranasal administration of glucagon, 1 mg, plus sodium glycocholate 15 mg as a surfactant, raised blood glucose levels and plasma levels of immunoreactive glucagon (IRG) and immunoreactive insulin (IRI). Spray solutions were more effective than drops, and neither the surfactant alone nor glucagon alone had any effect. Blood glucose levels were similarly affected by intravenous glucagon, while intramuscular glucagon was slightly more effective. The highest IRG concentrations were reached after intravenous administration, while intramuscular injection of glucagon was accompanied by the highest IRI release. These data indicate that intranasal administration of glucagon exerts metabolic effects similar to intramuscular and intravenous administrations. Further studies are needed to improve bioavailability and efficacy of intranasally administered glucagon.
Pontiroli, A. E., A. Calderara, et al. (1989).
"Intranasal glucagon as remedy for hypoglycemia. Studies in healthy
subjects and type I diabetic patients." Diabetes Care 12(9):
604-8.
Intranasal glucagon can raise blood glucose levels in healthy subjects. The aims of this study were to 1) compare the hyperglycemic effect of intranasal and intramuscular glucagon in healthy subjects and type I (insulin-dependent) diabetes patients during euglycemic conditions and 2) test the efficacy of intranasal and intramuscular glucagon in counteracting hypoglycemic episodes in insulin-treated diabetic patients. Intranasal glucagon raised blood glucose levels in both healthy subjects and type I diabetic patients, the effect of intramuscular glucagon being similar for the first 30 min and higher thereafter. Intranasal glucagon was also quicker acting than oral glucose in healthy subjects. Intranasal glucagon raised blood glucose levels in patients with hypoglycemic episodes, although less effectively than intramuscular glucagon. These data indicate intranasal glucagon as a possible emergency remedy for self-medication in insulin-treated patients prone to hypoglycemic episodes.
Pontiroli, A. E., A. Calderara, et al. (1993).
"Pharmacokinetics of intranasal, intramuscular and intravenous glucagon
in healthy subjects and diabetic patients." Eur J Clin Pharmacol
45(6): 555-8.
The pharmacokinetics of intranasal, an intravenous infusion, and intramuscular glucagon has been studied in 5 healthy subjects and 11 patients with insulin-dependent diabetes mellitus. After infusion the elimination half-life was significantly longer in diabetics (11.9 vs 6.6 min) and the apparent volume of distribution was twice as high in diabetics (0.19 vs 0.37 l.kg-1). The metabolic clearance rates were the same in the two groups (18.9 and 21.3 ml.min-1.kg-1 in controls and in diabetics) and were about twice those previously reported. After 1 mg intranasally the Cmax of immunoreactive glucagon (IRG) was similar in diabetic and in healthy subjects. Administration of a higher dose (2 mg) to diabetic patients produced a higher plasma level, although not proportionately so. The AUC after 1 mg was also similar in controls and in diabetics. The elimination half-life in both groups was similar to the value found after i.v. infusion; it was significantly shorter in controls (5.5 min) than in diabetics (13.8 min). In both groups, mean Cmax was significantly lower than after IM glucagon, the relative bioavailability of 1 mg intranasally vs IM injection being less than 30%. After IM administration, the Cmax and AUC of IRG in controls and in diabetic patients, were identical. The apparent elimination half-life was also similar in the two groups, and was three- to four-times longer (28.6 and 31.4 min) than after infusion or intranasal administration, possibly because estimation of the t1/2 was affected by slow release of the hormone from the site of injection.
Pontiroli, A. E., A. Calderara, et al. (1989).
"Intranasal drug delivery. Potential advantages and limitations from a
clinical pharmacokinetic perspective." Clin Pharmacokinet 17(5):
299-307.
Pontiroli, A. E. and G. Pozza (1990).
"Intranasal administration of peptide hormones: factors affecting
transmucosal absorption." Diabet Med 7(9): 770-4.
Pontiroli, A. E. (2015).
"Intranasal glucagon: a promising approach for treatment of severe
hypoglycemia." J Diabetes Sci Technol
9(1): 38-43.
Prevention of diabetic complications is mainly obtained through optimal
control of blood glucose levels. With hypoglycemic drugs like beta-cell
stimulating drugs and especially insulin, the limit to treatment is
represented by hypoglycemia, a life-threatening occurrence that is
dangerous itself and can induce fear of other episodes. Glucagon,
injected subcutaneously (SC) or intramuscularly (IM), is the treatment
of choice for severe hypoglycemia outside of the hospital setting.
However, due to practical aspects such as preparation of solutions for
administration and injection by untrained persons, there are obstacles
to its routine use. This review focuses on the current status of
alternative routes of administration of peptide hormones, and in
particular the intranasal (IN) route of glucagon, as a promising
approach for the treatment of severe hypoglycemia.
Pontiroli, A. E. and E. Tagliabue
(2020). "Intranasal versus injectable glucagon for hypoglycemia in type
1 diabetes: systematic review and meta-analysis." Acta Diabetol
57(6): 743-749.
AIMS: Glucagon is used to resolve severe hypoglycemia in unconscious patients with diabetes, requiring third-party assistance. A few studies have shown that intranasal (IN) glucagon causes resolution of hypoglycemia in insulin-treated patients with type 1 (T1DM) diabetes. This systematic review and meta-analysis updates the comparison of the effectiveness of IN glucagon with injected intramuscular/subcutaneous (IM/SC) glucagon in treatment of hypoglycemia in T1DM. METHODS: Controlled randomized studies were considered; eight studies, published in English, were included in a meta-analysis (random-effects model). Intervention effect (resolution of hypoglycemia) was expressed as odds ratio (OR), with 95% confidence intervals. Meta-regression was employed to correlate the effect with size of studies, age of patients, basal blood glucose levels. RESULTS: In a total of 467 treatments in 269 patients with IN and IM/SC glucagon, the OR IN versus IM/SC was 0.61 (CI 0.13-2.82); since four of eight studies showed 100% effectiveness, a simulation was made with 1 failure for each treatment; in this simulation analysis, the OR was 0.80 (95% CI 0.28-2.32). Heterogeneity was low and not statistically significant. Publication bias was absent, and quality of papers was high. At meta-regression, no correlation was found between the effect and number of patients in each study, age of patients, basal blood glucose levels. No study formally compared IN versus IM/SC in unconscious patients. CONCLUSIONS: This meta-analysis indicates that in conscious T1DM patients IN glucagon and IM/SC glucagon are equally effective in resolution of hypoglycemia.
Rickels, M. R., K. J. Ruedy, et
al. (2015). "Intranasal Glucagon for Treatment of Insulin-Induced
Hypoglycemia in Adults With Type 1 Diabetes: A Randomized Crossover
Noninferiority Study."
Diabetes Care.
Rosenfalck, A. M., I. Bendtson, et al. (1992).
"Nasal glucagon in the treatment of hypoglycaemia in type 1
(insulin-dependent) diabetic patients." Diabetes Res Clin Pract
17(1): 43-50.
The aim of this study was to compare the effect of nasally administered glucagon in doses of 1 (A) and 2 mg (B), with 1 mg glucagon administered intramuscularly (C) in 12 C-peptide-negative IDDM patients. Spontaneous recovery (D) from insulin-induced hypoglycaemia in the same patients was used as reference. The mean age was 31.1 (21-48) years, diabetes duration 10.8 (2.7-31) years and HbA1c 7.7 (6.5-9.8)%. Hypoglycaemia was induced by i.v. insulin infusion. When blood glucose (BG) reached about 2 mmol/l either glucagon was administered or the patients recovered spontaneously. BG nadir was 1.6 (1.1-2.3) mmol/l. BG increments during the first 15 min after glucagon administration were: (A) 1.9 +/- 0.7 (0.4-3.0); (B) 2.5 +/- 0.7 (1.5-3.5); (C) 2.5 +/- 1.0 (1.2-4.7); and (D) 0.3 +/- 0.4 (0-1.0) mmol/l, respectively. All treatments were more effective, measured as increments in BG, than spontaneous recovery, P less than 0.00001. There was no difference between nasal treatment with 2 mg (B) and i.m. treatment (C), both being more effective than 1 mg (A) nasal treatment, P less than 0.1. BG continued to increase up to 10 mmol/l 90 min after i.m. glucagon administration, whereas it stabilized at a level of 4.6-6 mmol/l, 30-45 min after nasal administration. Eighty percent of the patients had side-effects to nasal administration - local irritation, rhinitis or sneezing. Half of the patients sneezed, without correlation with the delivered dose of glucagon. None of the patients had side-effects which would preclude further treatment.(ABSTRACT TRUNCATED AT 250 WORDS)
Settles, J. A., G. F. Gerety, et
al. (2020). "Nasal Glucagon Delivery Is More Successful Than Injectable
Delivery: A Simulated Severe Hypoglycemia Rescue." Endocr Pract
26(4): 407-415.
Objective: A severe hypoglycemia (SH) episode is an acute, high-stress moment for the caregivers of persons with diabetes (PWD). We compared the success rates of nasal glucagon (NG) and injectable glucagon (IG) administration for PWD-trained and untrained users in treating simulated SH episodes. Methods: Thirty-two PWD-trained users and 33 untrained users administered NG and IG to high-fidelity manikins simulating treatment of an SH emergency. Simulation rooms resembled common locations with typical diabetic supplies and stressor elements mimicking real-life SH environments. Success rate and time to administer glucagon were measured. Results: Of all the PWD-trained and untrained users, 58/64 (90.6%) could successfully deliver NG, while 5/63 (7.9%) could successfully deliver IG. For NG simulations, 28/31 (90.3%) PWD-trained users and 30/33 (90.9%) untrained users could successfully administer the dose (mean time 47.3 seconds and 44.5 seconds, respectively). For IG simulations, 5/32 (15.6%) PWD-trained users successfully injected IG (mean time 81.8 seconds), whereas none (0/31 [0%]) of the untrained users were successful. Reasons for unsuccessful administration of NG included oral administration and incomplete pushing of the device plunger. For IG, inability to perform reconstitution steps, partial dose delivery, and injection at an inappropriate site were the causes for failure. Conclusion: With or without training, the success rate for administering NG was 90.6%, whereas it was only 7.9% for IG. NG was easily and quickly administered even by untrained users, whereas training was necessary for successful administration of IG. NG may expand the community of caregivers who can help PWD during an SH episode. Abbreviations: IG = injectable glucagon; NG = nasal glucagon; PWD = person with diabetes; SH = severe hypoglycemia; T1D = type 1 diabetes; T2D = type 2 diabetes.
Sibley, T., R. Jacobsen, et al. (2012). "Successful
Administration of Intranasal Glucagon in the Out-of-Hospital
Environment." Prehosp Emerg Care.
Abstract We present a case of successful prehospital treatment of
hypoglycemia with intranasal (IN) glucagon. Episodes of hypoglycemia can
be of varying severity and often requires quick reversal to prevent
alteration in mental status or hypoglycemic coma. Glucagon has been
shown to be as effective as glucose for the treatment of hypoglycemia.
The inability to obtain intravenous (IV) access often impairs delivery
of this peptide and is therefore frequently given via the intramuscular
(IM) route. Intranasal administration of glucagon has been shown to be
as effective as the IV route and may be used for rapid correction of
hypoglycemic episodes where IV access is difficult or unavailable and IM
administration is undesirable. We describe the first documentation in
the peer-reviewed literature of the successful treatment and reversal of
an insulin-induced hypoglycemic episode with IN glucagon in the
prehospital setting. We also present a review of the literature
regarding this novel medication administration route.
Singh-Franco, D., C. Moreau, et
al. (2020). "Efficacy and Usability of Intranasal Glucagon for the
Management of Hypoglycemia in Patients With Diabetes: A Systematic
Review." Clin Ther 42(9): e177-e208.
PURPOSE: Hypoglycemia is a common and sometimes life-threatening adverse event associated with insulin, sulfonylurea, and meglitinide therapies. In patients who are disoriented or unconscious, treatment with injectable glucagon is recommended, along with a call for emergency medical assistance. However, limitations of this formulation include difficulty with reconstitution and an unwillingness to administer an injection. In July 2019, intranasal glucagon was approved for use in the acute treatment of severe hypoglycemia in patients >/=4 years of age with diabetes. The purpose of this systematic review was to describe the efficacy, usability, and tolerability of intranasal glucagon 3 mg in patients with diabetes. METHODS: To identify studies, the following databases were systematically searched: Ovid MEDLINE, Embase, CINAHL, Web of Science Core Collection, Cochrane CENTRAL (EBSCO), ClinicalTrials.gov, and the World Health Organization International Clinical Trials Registry Platform, from inception to March 3, 2020. Comparative studies included patients with diabetes and an active comparator. Usability studies enrolled participants who used a device for glucagon administration. FINDINGS: Ten studies met the inclusion criteria. In 5 comparative studies in insulin-induced hypoglycemia (intranasal vs injectable glucagon), the criteria for successful treatment varied. In 3 studies, it was defined as an increase in blood glucose of >/=70 mg/dL (3.9 mmol/L) or an increase of >/=20 mg/dL (1.1 mmol/L) within 30 min of glucagon administration. In 1 study, the criteria were stricter, with success defined as an increase in blood glucose of >/=27 mg/dL (>/=1.5 mmol/L) within 15 min. In the pediatrics study, success was defined as an increase in blood glucose of >/=25 mg/dL (1.4 mmol/L) within 20 min. In 2 studies of intranasal glucagon monotherapy in clinical practice, the primary end point was the percentage of patients who awakened or returned to normal status within 30 min of intranasal glucagon administration. In these 7 studies, almost all of the participants met the criteria for success as defined in their respective studies. The mean time to treatment success was between 10 and 20 min with intranasal and injectable glucagon. Nausea and vomiting were common adverse events with both formulations; watery eyes and runny nose occurred more frequently with intranasal glucagon. In 3 simulation studies, caregivers and noncaregivers administered intranasal glucagon within 1 min versus 1.3-5 min with IM glucagon. IMPLICATIONS: In patients who are disoriented or unconscious, treatment with injectable or ready-to-use intranasal glucagon increases blood glucose within 15-30 min. Intranasal glucagon was preferred by most caregivers and noncaregivers due to its ease of use. Additional studies of intranasal glucagon in younger patients (1-<3 years of age), pregnant women, and in comparison with SC glucagon are needed to further clarify bioavailability, efficacy, and tolerability.
Sherr, J. L., K. J. Ruedy, et al. (2016). "Glucagon Nasal Powder: A
Promising Alternative to Intramuscular Glucagon in Youth With Type 1
Diabetes." Diabetes Care
39(4): 555-562.
OBJECTIVE: Treatment of severe hypoglycemia outside of the hospital setting
is limited to intramuscular glucagon requiring reconstitution prior to
injection. The current study examined the safety and dose-response
relationships of a needle-free intranasal glucagon preparation in youth
aged 4 to <17 years. RESEARCH DESIGN AND METHODS: A total of 48 youth
with type 1 diabetes completed the study at seven clinical centers.
Participants in the two youngest cohorts (4 to <8 and 8 to <12 years
old) were randomly assigned to receive either 2 or 3 mg intranasal
glucagon in two separate sessions or to receive a single, weight-based
dose of intramuscular glucagon. Participants aged 12 to <17 years
received 1 mg intramuscular glucagon in one session and 3 mg intranasal
glucagon in the other session. Glucagon was given after glucose was
lowered to <80 mg/dL (mean nadir ranged between 67 and 75 mg/dL).
RESULTS: All 24 intramuscular and 58 of the 59 intranasal doses produced
a >/=25 mg/dL rise in glucose from nadir within 20 min of dosing. Times
to peak plasma glucose and glucagon levels were similar under both
intramuscular and intranasal conditions. Transient nausea occurred in
67% of intramuscular sessions versus 42% of intranasal sessions (P =
0.05); the efficacy and safety of the 2- and 3-mg intranasal doses were
similar in the youngest cohorts. CONCLUSIONS: Results of this phase 1,
pharmacokinetic, and pharmacodynamic study support the potential
efficacy of a needle-free glucagon nasal powder delivery system for
treatment of hypoglycemia in youth with type 1 diabetes. Given the
similar frequency and transient nature of adverse effects of the 2- and
3-mg intranasal doses in the two youngest cohorts, a single 3-mg
intranasal dose appears to be appropriate for use across the entire 4-
to <17-year age range.
Slama, G., C. Alamowitch, et al. (1990). "A new
non-invasive method for treating insulin-reaction: intranasal
lyophylized glucagon." Diabetologia 33(11): 671-4.
The main therapeutic indication for glucagon is the treatment of hypoglycaemia in insulin overdosed Type 1 (insulin-dependent) diabetic patients. We have previously shown that an intranasal spray of 7.5 mg glucagon with deoxycholic acid as surfactant was able to correct an i.v. insulin-induced hypoglycaemia in diabetic patients. However, bioavailability and stability needed to be improved before intranasal glucagon could be introduced into clinical practice. This has now been achieved with a freeze-dried mixture of glucagon (1 mg) and glycocholic acid (1 mg) as a surfactant. Kinetics and efficacy have been controlled by (1) comparing subcutaneous and intranasal glucagon in 12 healthy non-hypoglycaemic subjects; (2) testing intranasal glucagon in six Type 1 diabetic patients in whom hypoglycaemia was induced by an i.v. bolus of insulin and (3) comparing subcutaneous and intranasal glucagon in six Type 1 diabetic patients in whom hypoglycaemia was induced by adding extra subcutaneous regular insulin to their usual morning dosage. Our results show that 1 mg of intranasal glucagon is as effective as 1 mg of subcutaneous glucagon in terms of the rise in blood glucose. Differences in kinetics between the subcutaneous and the intranasal routes may be observed: intranasal glucagon initiates the blood glucose rise earlier than does the subcutaneous form but the effect of the latter is more sustained. Glycocholic acid appears to be a perfectly tolerated agent in acute conditions. The use of intranasal lyophylized glucagon, for the reversal of hypoglycaemia in Type 1 diabetes, seems to be a clinically relevant alternative to its parenteral equivalent and should now be ready to be introduced in the market.
Slama, G., L. Freychet, et al. (1988).
"Intranasal glucagon for hypoglycaemia [letter]." Lancet 2(8614):
799.
Slama, G., G. Reach, et al. (1992). "Intranasal
glucagon in the treatment of hypoglycaemic attacks in children:
experience at a summer camp." Diabetologia 35(4): 398.
Stenninger, E. and J. Aman (1993). "Intranasal glucagon treatment relieves hypoglycaemia in children with type 1 (insulin-dependent) diabetes mellitus." Diabetologia 36(10): 931-5.
The aim of the present study was to compare intra-nasal glucagon with subcutaneous glucagon as a treatment of insulin-induced hypoglycaemia in 11 children, 7-12 years old, with Type 1 (insulin-dependent) diabetes mellitus. Hypoglycaemia (1.6 +/- 0.1 vs 1.8 +/- 0.2 mmol/l) was induced twice in each child by continuous insulin and variable glucose infusions. One milligram of intranasal glucagon or 0.5 mg of subcutaneous glucagon was given in a randomized order. At 15 min after the administrations of either intranasal or subcutaneous glucagon, the blood glucose concentration increased by 1.5 +/- 0.2 mmol/l or 1.7 +/- 0.2 mmol/l above the glucose nadir, respectively. After nasal administration, the maximal rise in blood glucose was seen after 25 min. Subcutaneous injections induced higher and more sustained plasma glucagon concentrations but the children suffered more often from nausea than when they were treated intranasally. In conclusion, treatment with intranasal glucagon seems to be efficient and results in a rapid correction of insulin-induced hypoglycaemia with few side-effects.
Suico, J. G., U. Hovelmann, et
al. (2020). "Glucagon Administration by Nasal and Intramuscular Routes
in Adults With Type 1 Diabetes During Insulin-Induced Hypoglycaemia: A
Randomised, Open-Label, Crossover Study." Diabetes Ther 11(7):
1591-1603.
INTRODUCTION: Many commercially available glucagon products for
treatment of severe hypoglycaemia require cumbersome reconstitution and
potentially intimidating injection during an emergency. Nasal glucagon
(NG) is a novel drug-device combination product consisting of a
single-use dosing device that delivers glucagon dry powder through nasal
administration. The present study assessed whether 3 mg NG was
non-inferior to 1 mg intramuscular glucagon (IMG) in adults with type 1
diabetes. METHODS: This randomised, open-label, two-period, crossover
trial was conducted at two clinical sites. Hypoglycaemia (plasma glucose
[PG] target of < 3.3 mmol/l (60 mg/dl) was induced by an intravenous
insulin infusion. Glucagon preparations were given by study staff.
Treatment success was defined as an increase in PG to >/= 3.9 mmol/l (70
mg/dl) or an increase of >/= 1.1 mmol/l (20 mg/dl) from the PG nadir
within 30 min of receiving glucagon. RESULTS: Of the 66 participants
included in the primary efficacy analysis who received both NG and IMG,
100% achieved treatment success, thus demonstrating non-inferiority of
NG to IMG. All participants achieved treatment success within 25 min
with the mean time to treatment success of 11.4 min (NG) and 9.9 min
(IMG). No serious adverse events occurred. Forty-eight
treatment-emergent adverse events (TEAEs) occurred after NG and 51 after
IMG. Most TEAEs were mild and transient. CONCLUSION: Nasal glucagon was
as efficacious and well tolerated as IMG for the treatment of
insulin-induced hypoglycaemia in adults and will be as useful as IMG as
a rescue treatment for severe hypoglycaemia. TRIAL REGISTRATION:
NCT03339453, ClinicalTrials.gov.
Teshima, D., A. Yamauchi, et al. (2002). "Nasal glucagon delivery using microcrystalline cellulose in healthy volunteers." Int J Pharm 233(1-2): 61-6.
We developed an intranasal powder form of glucagon to improve metabolic status and fatty liver in patients with pancreatectomy. Microcrystalline cellulose, which is commonly used in commercial preparations for allergic rhinitis was used as an absorption enhancer. We compared the intranasal powder form with some spray solutions of glucagon with regard to glucagon absorption, concentration of blood glucose, stability and nasal irritation. The absorption of glucagon from the spray solution including 1.5% sodium glycocholate or 1% sodium caprate was 1.3- and 2.6-fold higher than that from the powder form mixed with microcrystalline cellulose at a ratio of 1:69, respectively. The C(max) values of plasma glucose were 2.18, 3.39 and 1.56 mmol l(-1) in the spray solutions including sodium glycocholate and sodium caprate and in the powder form, respectively. However, glucagon in spray solutions was unstable, but that in the powder form was stable at 5 and 25 degrees C for at least 84 days. The spray solution caused strong irritation, but the powder form did not. These results suggested usefulness of the powder form of glucagon for treatment of pancreatectomized patients.
Ueno, H., M. Mizuta, et al. (2014). "Exploratory trial of intranasal
administration of glucagon-like peptide-1 in Japanese patients with type
2 diabetes." Diabetes Care
37(7): 2024-2027.
OBJECTIVE: This study aimed to assess the efficacy and safety of our newly developed nasal glucagon-like peptide-1 (GLP-1) compound and injector. RESEARCH DESIGN AND METHODS: Twenty-six patients with type 2 diabetes were enrolled in this double-blind placebo-controlled study. The nasal compound containing 1.2 mg of human GLP-1 (7-36) amide or placebo was administered immediately before every meal for 2 weeks. RESULTS: The plasma peak concentration of active GLP-1 was 47.2 pmol/L, and its Tmax was 8.1 min. The early phase of insulin and glucagon secretion were recovered and suppressed, respectively, in the GLP-1 group. Glycoalbumin levels became significantly lower and 1,5-anhydroglucitol levels significantly higher after GLP-1 administration. No marked adverse events were observed after using nasal GLP-1. CONCLUSIONS: The newly developed nasal GLP-1 compound may be a potential treatment for type 2 diabetes. The long-term application of the drug should be evaluated in future trials.
Yanai, O., M. Phillip, et al. (2005). "IDDM patients' opinions on the use of glucagon emergency kit in severe episodes of hypoglycemia." Practical Diabetes 14(2): 40-42.
This study investigated the coping strategies of IDDM patients with mild and severe episodes of hypoglycaemia. One hundred and two IDDM patients, aged 4-62 years old, 59% male and 41% female, were interviewed by telephone using a structured questionnaire. Most of the patients recognised the symptoms of hypoglycaemia and accordingly treated their mild episodes with either Coca Cola or fruit juice (61%), sweets (32%), milk (26%), or a slice of bread (23%). Forty two per cent of the patients treated themselves with additional chocolate, fruit or honey. While 83% of the patients reported that they had received an explanation regarding the use of the glucagon emergency kit, only 60% actually owned it. In only 19% of the patients who had ever experienced a severe episode of hypoglycaemia had this mode of treatment actually been used. Most of the patients (67%) said they would prefer the intranasal route if available and 82% assumed that the people surrounding them would prefer to administer an intranasal spray or drops in emergency hypoglycaemic situations. We conclude that only in a small percentage of patients is subcutaneous or intramuscular glucagon used in an emergency situation, and speculate that other modes of glucagon administration such as the intranasal route would increase the use of glucagon and prevent some of the IDDM patients from waiting for the emergency paramedic teams or from being referred to hospitals.
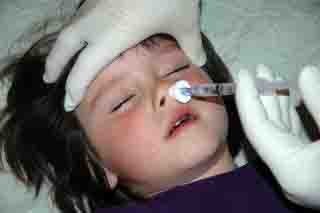 Therapeutic
Intranasal Drug Delivery
Therapeutic
Intranasal Drug Delivery