Intranasal midazolam for acute seizure therapy
Table of contents:
Introduction to IN medications for seizures (Click here)
Literature overview and discussion (Click here)
Personal insights from experienced clinicians (Click here)
Treatment protocol (Click here)
Midazolam Dosing chart (Click here)
Teaching materials (Click here)
Introduction
Breakthrough
seizures are all too commonplace in patients with epilepsy.
Some of these seizures will prove to be prolonged.
There are an estimated 150,000 cases of status epilepticus every
year in the US.[1] Caretakers, prehospital and hospital providers are
frequently faced with management of prolonged seizure activity.
As morbidity and mortality are at least partially dependent on
the duration of seizure activity it is crucial that seizures be stopped
as soon as possible.
Transmucosal delivery of benzodiazepines (midazolam or lorazepam)
provides a very effective, safe and inexpensive means to rapidly achieve
seizure control.
Seizures are to a
certain degree self-sustaining.
Because global central nervous system activity is a balance of
excitatory and inhibitory neurotransmission factors that shift the
balance toward excitation can lead to prolonged seizure activity.
Many clinicians have noted through their practice experience that
seizures seem to be self-reinforcing.
A molecular explanation for this phenomenon is beginning to
develop. Mounting evidence
suggests that post-synaptic GABAA receptors are internalized
during status epilepticus thereby reducing neuro-inhibition at the
synapse.[2] The principle first-line agents for the treatment of status
epilepticus are the benzodiazepines, which are GABA agonists.
Therefore the longer a seizure persists the less effective this
class of medicines may be suggesting that the earlier benzodiazepines
are delivered the better.
Transmucosal
delivery of generic benzodiazepines via the nasal mucosa offers an
attractive and cost-effective alternative in the out-of-hospital
setting. Midazolam and lorazepam easily cross the nasal mucosa and the
blood brain barrier, resulting in a rapid rise in both the plasma and
the cerebrospinal fluid concentrations.[3-6, 27]
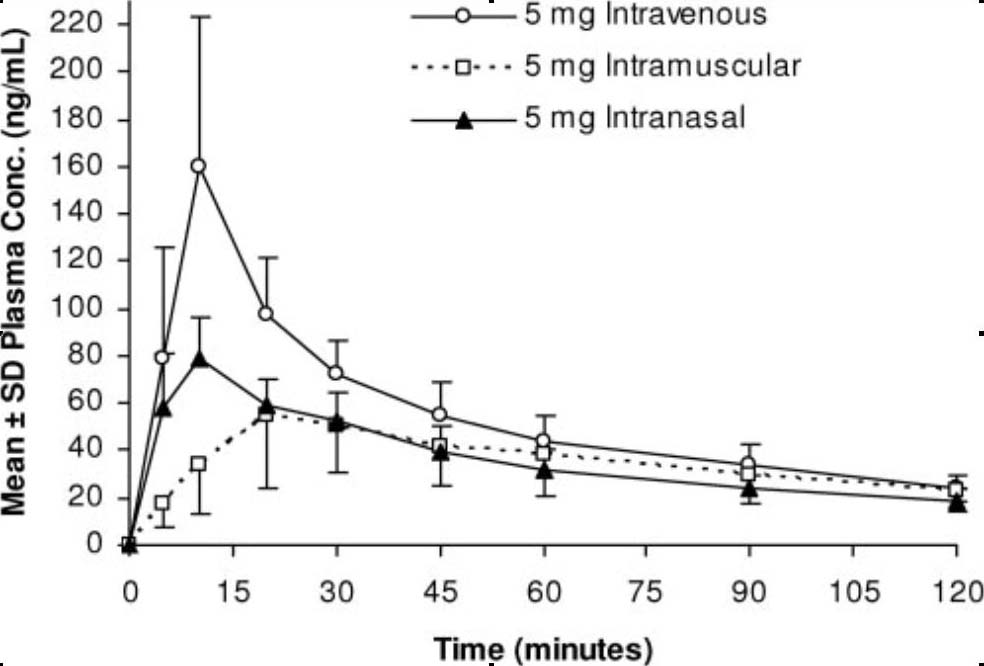
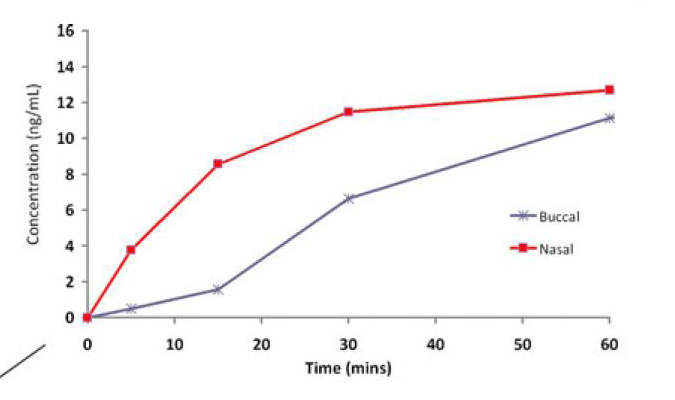
Wermeling et al.
(2006) Pharmacokinetics and pharmacodynamics of a new intranasal
midazolam formulation (25 mg/ml concentration) in healthy volunteers.[6]
Literature overview and discussion
Intranasal midazolam
Three randomized
controlled trials and 1 prehospital observational trial exist, comparing
rectal diazepam to either buccal (oral transmucosal) or intranasal
midazolam.[7-10] Scott et al
conducted a randomized controlled trial comparing buccal midazolam to
rectal diazepam in epileptic students in an extended care school.[7]
A school nurse administered medication to all students who
suffered continuous seizures for more than a 5-minutes. Patients with
persistent seizures for an additional 10 minutes were treated at the
on-call physician’s discretion. Oral transmucosal midazolam was
effective in 75% of cases (30 of 40 seizures), whereas rectal diazepam
was effective in 59% (23/39) ( P = non significant). There were no
adverse cardiorespiratory effects in either group. Although these
differences did not achieve statistical significance, the trend toward a
better outcome along with the more socially acceptable delivery of oral
transmucosal medication led the school to change its preferred treatment
to the oral transmucosal route.
Camfield et al
found
similar efficacy in their randomized trail comparing these 2 routes and
drew identical conclusions—oral transmucosal midazolam was preferred
over rectal diazepam because of ease of use and social acceptability.[8]
The third randomized controlled trial, conducted by Fisgin et al,
compared intranasal (rather than buccal) transmucosal midazolam to
rectal diazepam.[9] In this study, midazolam aborted 20 (87%) of 23
seizures and rectal diazepam 13 (60%) of 22 seizures (P b .05). These
results were statistically significant in favor of the intranasal 130
route when compared with the rectal route. Again, as in previous
studies, no clinically important adverse events were identified in the 2
groups.
The final study,
by Holsti et al, was conducted in a prehospital ambulance setting.[10]
In this study, the entire emergency medical system converted from rectal
diazepam to intranasal midazolam for treatment of pediatric seizures.
The authors compared effectiveness and complication data before and
after the change. The rates of prehospital seizure control (100% vs.
78%), need for need for emergent intubation (0% vs. 33%), and need for
hospital admission (40% vs. 89%) were all substantially less in the
intranasal midazolam group compared with the rectal diazepam group. The
mean seizure duration was 11 minutes for those treated with nasal
midazolam compared to 30 minutes for rectal diazepam.
Children given the rectal medication were significantly more
likely to have another seizure in the ED (odds ratio [OR] 8.4), need ED
intubation (OR 12.2), need hospital admission (OR 29.3) and need ICU
admission (OR 53.5).
All these authors
conclude that trans-mucosal midazolam is more convenient, easier to use,
just as safe, and is more socially acceptable than rectal diazepam.
Furthermore, when given via the intranasal route, midazolam is more
effective than rectal diazepam.
The above evidence
clearly suggests that intranasal midazolam is superior to rectal
midazolam for seizure therapy in children. However, IV benzodiazepines
are first-line therapy in most hospitals—how does intranasal midazolam
compare to IV benzodiazepines? Three randomized controlled trials
comparing intranasal midazolam to IV diazepam answer this question.[11,
12, 26] Lahat et al compared intranasal midazolam to IV diazepam in children
seizing 10 minutes or longer.[11] Patients were randomized to receive
diazepam, 0.3mg/kg IV, or midazolam 0.2 mg/kg intranasally. Nasal
midazolam stopped 23 (88%) of 26,whereas 24 (92%) of 26 were controlled
with IV diazepam (P = non significant). The mean time from patient
arrival to seizure cessation was 6.1 minutes with midazolam and 8.0
minutes with diazepam. The authors conclude that intranasal midazolam
was as safe and effective as IV diazepam, but the overall time to
cessation of seizures after arrival at the hospital was faster with
intranasal midazolam because of the time required to establish an IV
line in the diazepam group. A similar study was conducted by Mahmoudian
and Zadeh.[12] These authors compared the efficacy of intranasal
midazolam (0.2 mg/kg) to IV diazepam (0.2 mg/kg) in 70 patients (ages 2
to 15 years) presenting to the emergency department with seizure
activity. Both methods were equally effective, and no adverse effects
occurred in either group. Javadzadeh compared IV diazepam 0.3 mg/kg to
intranasal midazolam 0.2 mg/kg in children suffering prolonged
seizures.[26] (click
here for free article) From time of drug delivery to cessation of
seizure IV drug worked in 2.16 minutes while nasal worked in 3.16
minutes. However, due to delays related to establishing an IV, the time
to control a seizure using IV diazepam was statistically and clinically
significantly longer (6.42 minutes). Both therapies were 100% effective
in the 60 patients treated. Thakker also conducted a randomized trial
comparing IN midazolam to IV diazepam showing clinical equivalency in
terms of seizure cessation but much faster resolution of the seizure
when the nasal drug is given – all due to no need for an IV line. The
time differences may be clinically significant in terms of brain hypoxia
and potential long term outcomes – 6.7 minutes with nasal drug versus
17.2 minutes with IV therapy. In this study the resolution rates for
single dose therapy were only 65 to 66% - very similar to other studies
conducted outside of wealthy western countries (this study is from
India). This is probably due to the very high incidence of CNS
infections which was 26% in this study.
Sharma and Harish provide yet another
prospective study on the topic in 2013.[31] They randomized 100 children
in their ER or NICU who were seizing to either intranasal midazolam or
intravenous midazolam for the treatment of status epilepticus. As in
every other prospective trial on the topic the two treatment are
equivalent in terms of efficacy: In this study both were successful at
stopping seizures 94% of the time with a single dose. The intranasal
route was also slightly faster likely due to the delays associated with
IV line establishment. Unique to this study was the inclusion of
neonates: There were 7 neonates in this study and an additional
26 children under the age of 1 year. (Click
here for free article)
All these authors conclude that nasal midazolam
works faster, is safe, is easy to use and
should be used not only in medical centers but also in general
practitioners’ offices as well as at home by families of seizure-prone
children after appropriate instruction.
Perhaps the
greatest benefit of intranasal midazolam will be for the treatment of
seizures in the prehospital, home or extended care setting. Wilson et al sent intranasal midazolam home with families of
children suffering epilepsy and found that 33 of 40 (83%) who used it
found it effective and 20/24 (83%) preferred using transmucosal
midazolam to rectal diazepam.[13] Harbord reported experience using
intranasal midazolam for home treatment of 54 seizures in 22
children.[14] These authors found it to be 89% effective, with no
evidence of respiratory compromise. Ninety percent of families found no
difficulty with nasal medication administration. Of the 15 parents with
previous rectal diazepam experience, 13 thought intranasal delivery was
easier and 14 preferred it to the rectal route. Jeannet et al, used
intranasal midazolam both on the medical wards and as home therapy.[15]
Their experience with 26 children suffering 125 seizures note a 98%
effectiveness in under 10 minutes with no serious adverse effects.
When compared to rectal diazepam they report that the intranasal
route was both easier to use and that postictal recovery was faster.
Scheepers et al report their experience with intranasal medication
delivery in an extended care facility caring for adolescents and adults
with severe epileptic disorders.[16] Of 84 uses, they found this route
to be effective in 79 (94%). In the 5 instances when it was not
effective, 3 of the 5 doses were delivered intra-orally rather than
intranasally.
Click here for the PDF downloaded from the free thesis link
Also in 2013, Humphries and Eiland wrote a review of intranasal
midazolam for the treatment of acute seizures.[30] The article provides
a very nice up to date overview of the pharmacokinetics and absorptive
properties of benzodiazepines for anyone who is interested. They also
provide an extensive review of the literature regarding the two
currently available rapid methods of treating seizures in an outpatient
setting – rectal diazepam versus nasal midazolam – including cost
differences (a few hundred dollars for ONE DOSE
for rectal versus less than 20 dollars a dose for nasal). They conclude
that “Intranasal midazolam should be considered as an anticonvulsant
agent for community, prehospital, and ED use in children when
intravenous access is not available and the rectal route is not
desirable.”
(Click
here for link to FREE ARTICLE)
In 2015, Bancke et al found that a commercially
reformulated form of IN midazolam
shows superior bioavailability, though this does not necessarily
translate into improved clinical outcomes nor does it imply
affordability
Crawford et al provide an overview of IN midazolam use and ideas for
implementation in a community practice.[43] The article is packed full
of data that essentially mirrors what is stated in this web site: When
comparing IN midazolam to rectal diazepam for outpatient treatment of
status epilepticus the IN route is more effective, costs much less,
results in better outcomes (shorter seizure time, less admission to
hospital), has less of a social stigma and is markedly preferred by
families of the epileptic patient. Based on this information the authors
attempted to change practice in their medical community and transition
their neurologic practice over to prescribing IN midazolam as the
preferred outpatient rescue medication and make it available from local
pharmacies. They note very rapid adoption in the practice after
presenting detailed information and setting up local pharmacies with
delivery devices and appropriate drugs. Further challenges involved
school nurse education when children showed up with a new drug delivery
system. This paper addresses this and many challenges and should be read
by any clinician who anticipates adopting IN benzodiazepine use in their
community.
Nunley et al compared healthcare utilization rates in 5458 children who
were using either intranasal midazolam or rectal diazepam (or both) to
treat acute seizures. They found a significant reduction in ED visits,
urgent care visits and ambulance transports in children using IN
midazolam over those using rectal diazepam. They conclude that their
results “substantiate the cost-effective benefits of prescribing
intranasal midazolam compared to per rectum diazepam”.
Crawshaw and Cock have put transmucosal benzodiazepines into their
status epilepticus algorithm as the first line therapy in patients
without an IV stating “Speed of administration and adequate initial
dosing are probably more important than the choice of drug.” [59]
(Free access on internet – click here for PDF)
The EMS system in Zurich Switzerland had a shortage of IV lorazepam so they
tested IN midazolam single dose (the dose is unknown to this writer) and
IV diazepam on their adult seizure patients. They found IN midazolam in
one dose to be effective in terminating seizures in 57% of adults
suffering status epilepticus – good first line results in patients
without an IV but not ideal or as high as the 85-90% seen in children.
Intranasal Lorazepam
Intranasal
lorazepam, another anti-seizure medication, also appears effective and
safe for treating status epilepsy based on a single trial. A large study conducted in rural Africa compared intranasal
lorazepam to intramuscular paraldehyde (a common medication used in the
third world to seizure therapy) in 160 patients, most of whom were
seizing for an extended time period (mean seizure duration was over 2
hours).[17] Despite these prolonged seizures, intranasal lorazepam
managed to stop three quarters of the seizures in a few minutes, while
intramuscular medication only worked 61% of the time. There were no
respiratory side effects seen with the lorazepam.
However, by 2015 it appeared they persevered, or at least took the data they had and published it in an African medical journal:
Lissauer et al
conducted a prospective open label randomized trial in
African children suffering status epilepsy to determine whether
lorazepam (0.1 mg/kg) given via the IV, IN or buccal route was
superior.[38] They found seizures were controlled within 10 minutes in
46% of the buccal group, 57% of the nasal group and 83% of the IV group.
These results are not surprising when one looks at their patient
population: Over 10% had HIV,
over 50% had malaria, many seizures lasted hours prior to treatment,
most patients did not have idiopathic epilepsy (<17%), over 25% had
cerebral malaria and an additional 15-20% had bacterial meningitis,
15-20% died. As often
stated in this web site, nasal drug delivery is simply another tool for
delivering anti-seizure medications rapidly while IV therapy is a higher
“gold standard” in many clinical settings and should be established
based on patient severity of illness. Were
I to work in this clinical setting I would very rapidly move to
establishing an IV in any seizure patient seen here given the
populations illness severity. Unless you work in a setting where
seizures are a marker of such severe infectious illness, you cannot
extrapolate these results to your practice. These authors concur and
state that in their clinical setting, the IV route is superior but
transmucosal drug delivery (buccal, nasal) should be implemented when IV
access cannot be rapidly obtained.
Another study and its accompanying editorial of importance to this discussion were published in the New England Journal of Medicine in February 2012.[24, 25] While this study does not actually use intranasal medications to treat seizures, it is a very important and well done trial that is applicable to the discussion regarding therapy for status epilepsy. These authors found that intramuscular midazolam was as good if not better than intravenous lorazepam for rapid termination of status epilepsy. The reason is due to ease of drug delivery and therefore rapid drug delivery. Of course this is also the reason that intranasal midazolam and lorazepam are as effective as intravenous benzodiazepines in all the studies reviewed here. For some reason they make a very odd comment supported by absolutely no data - "Midazolam can be administered by other non-intravenous routes as well, but the intramuscular route is more consistently effective than the intranasal or buccal routes because the drug cannot be blown or spat out by the convulsing patient." This is not true. First of all, the literature suggests otherwise and the editorial in the same issue of the NEJM points out 3 reference showing IM benzodiazepines absorb LESS rapidly than nasal drugs (two references can be found in the seizure section of this web site - O'Regan 1996, Wermeling 2009). Secondly, this statement shows the lack of experience of the authors who wrote it - IN drugs are very easy to deliver to a seizing patient if you use a syringe driven atomizer and the patient is unconscious so they can't spit it back at your. Finally, many other studies for other drugs like opiates show more rapid onset of action and much higher patient satisfaction with IN versus IM drugs so I am not sure why these authors or the NEJM editorialist would allow this opinion to be stated. Never this less, this is a great study that demonstrates non intravenous therapy is essentially as effective as IV delivery of benzodiazepines for treatment of seizures and it is a lot easier to administer. It will be interesting to find out what the final version of this therapy costs. Currently the only FDA approved drug for out-patient seizure therapy is rectal diazepam and it costs well over $200-300 per dose in a U.S. pharmacy while being much less effective than nasal, intramuscular or intravenous therapy. Given this current cost, this new therapy will likely demand a similar price. Our EMS system, local emergency departments and our community pediatric neurologists use generic midazolam as first line therapy in the EMS, ER and outpatient setting - at about $2-3 per dose plus a few dollars for the delivery device. They have used this therapy for almost a decade and their success rates are similar or superior to those found in this study.[10,18]
Intranasal Diazepam
Although early data demonstrated poor bioavailability of nasal diazepam, the pharmaceutical industry has recognized an opportunity for nasal benzodiazepines and we are in the midst of research with modified diazepam molecules to make them more available via the nasal mucosa.[36] There is a plethora of older data on this but as of 2014 it appears to be more mature so it is time to begin providing recent literature on this topic.
Henney et al found a modified IN diazepam
formulation (enhanced for nasal delivery) similar in terms of
bioavailability as rectal diazepam gel. They believe this offers
caregivers and patients a more socially acceptable and convenient method
for treating seizures.
Inokuchi et al retrospectively compared their experience using
intranasal diazepam or intravenous diazepam for controlling status
epilepsy in acute stroke patients presenting to their institution.[37]
They found IN drug was delivered 9 times faster ( 1 vs. 9 minutes) and
seizure control occurred three times faster (3 versus 9.5 minutes). They
conclude “Intranasal diazepam administration is safer, easier, and
quicker than intravenous administration.”
Costs of care:
Caron et al conducted a study that quantified the CHARGES (not cost) for
pediatric seizure care of 90 patients who discharged home from a US
hospital emergency room.[40] The
average charge was $2444 with a range of $370 to $17126. The authors
state they provide this data so clinicians can understand the costs and
compare the values of various treatment options (like home rectal
diazepam or IN midazolam). (It should be noted that at the end of this
section of the website protocols with teaching materials are available
that provide you clinicians with a method
to provide effective and safe home care at about 12 dollars or
less per treatment and which the literature notes to be 90% effective –
thereby allowing the family to stay home and save the $2444 as well as
the ambulance charges).
Sanchez Fernandez utilized published data on seizure cessation with
various delivery methods (Nasal, buccal, IM, and rectal benzodiazepines)
and then obtained available market prices of each of these
medications.[49] They found that nasal and buccal midazolam are the most
cost effective methods and that rectal diazepam is never cost effective
given it lower efficacy and markedly higher price (50 times more
expensive and less effective). Interestingly, they point out that rectal
diazepam does NOT have regulatory approval for treating ongoing seizures
or status epilepticus. Instead it is approved just for reducing
frequency of seizures and its place in status epilepticus treatment
algorithms is off label and is very questionable given its poor
performance in these situations. For these reasons,
rectal diazepam is no longer
recommended for use in status epilepsy by the American epilepsy society
– rather they recommend non-IV midazolam delivery methods.
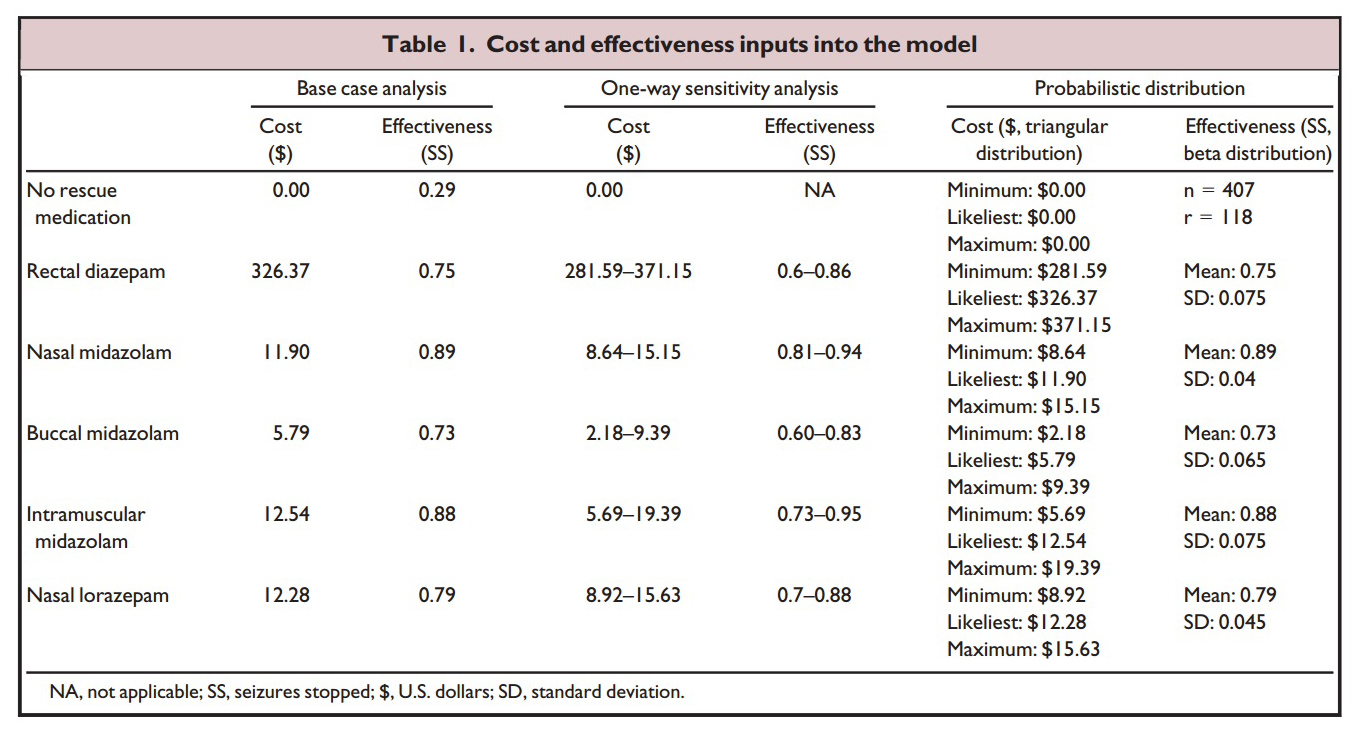
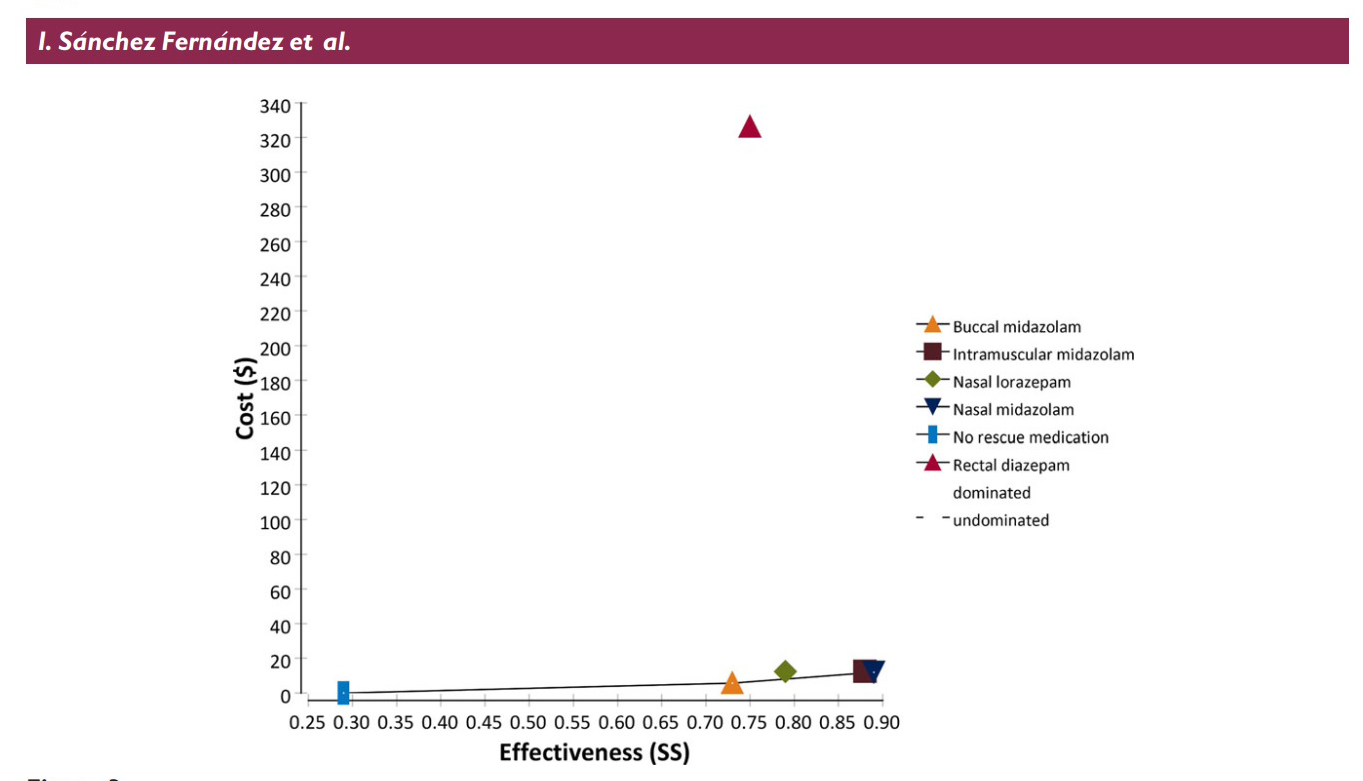
The table and graph above shows the parameters Sanchez Fernandez et al
used to determine their results: Cost is the total cost of the drug plus
applicator (nasal atomizer or syringe and needle etc). Effectiveness is
the literature reported percentage of cases (from 24 studies reviewed)
that the medication and delivery route will stop prolonged seizures. The
graph plots the cost versus the effectiveness with the ideal drug being
one that plots out at the lower right corner of the graph (low cost,
highly effective). As is readily apparent rectal diazepam is an absolute
outlier.
Conclusion:
This data is
fairly compelling. Multiple studies from different authors throughout
the world confirm that intranasal midazolam (and in now IN lorazepam)
is a safe and very effective means to achieve seizure control at home,
in the ambulance and in the hospital.
Its rapid onset of action, social acceptability, ease of use and
efficacy suggest that this therapy should be adopted in appropriate
situations (prolonged seizures) where IV access is not immediately
available.
Personal insights from experienced clinicians
Tom Macfarlane, MD. Emergency Physician, Salt Lake City, Utah:
I have extensive experience in using intranasal midazolam in the hospital and home settings. As an emergency physician I frequently care for patients who are experiencing seizures. It is often difficult to establish intravenous access in these seizing patients and therefore transmucosal drug delivery is a great option. I also have a 4 year-old son who experiences frequent prolonged seizures. My family and I have safely administered over 60 transmucosal doses of midazolam over the course of 3 years. This has allowed us to avoid many trips to the Emergency Department and provides us with a degree of increased freedom. I am confident that my son would not be functioning at the level he is today without intranasal midazolam.
Treatment protocol:
Note: Midazolam is now available in prefilled syringes. Just connect an atomizer and deliver the correct dose.
Prefilled syringes of medication:
Based on extensive research and community use, prepackaged midazolam in the 5mg/ml dosing would certainly seem appropriate for use by properly trained families and BLS providers - and this packaging would surely help improve the ease of use. See this PDF file for the information on many prepackaged medications including midazolam and lorazepam: Carpuject medications In situations where the entire volume might not be appropriate (smaller child), the syringe can have a indelible mark or piece of tape applied by the clinician or pharmacy at the appropriate dosage so the parent/provider knows how much to give. Just be sure to use the proper concentration.
Carpuject of midazolam (has a Luer attachment for a needle or atomizer)
Intranasal midazolam for acute seizures
Indications: For treatment of persistent seizure activity
Procedure:
- Assess ABC’s – Airway, Breathing, Circulation
- For pulseless patients, proceed to ACLS guidelines
- Apply 100% oxygen NRB mask to seizing patient
- Use age based table to determine proper volume of midazolam for atomization (see table below)
- To calculate it manually, use the below
formula
-
Assess
weight: children weight in kg = 10 + 2(Age in years)
-
Calculate appropriate dose of midazolam using the following
formula:
-
Children: Total kg wt X 0.2 mg = total mg dose of midazolam,
maximum of 10 mg
-
Adults over 50 kg: 10 mg (2 ml) of midazolam
- Total volume in milliliters of midazolam (5mg/ml concentration) = (Total mg dose divided by 5mg/ml) + 0.1 ml for dead space of device.
-
Children: Total kg wt X 0.2 mg = total mg dose of midazolam,
maximum of 10 mg
-
Assess
weight: children weight in kg = 10 + 2(Age in years)
- (If using lorazepam - 0.1 mg/kg to max 4 mg)
- Load syringe with appropriate milliliter volume of midazolam (use only 5mg/ml concentration) and attach nasal atomizer
- Place atomizer within the nostril
- Briskly compress syringe to administer 1/2 of the volume as atomized spray.
- Remove and repeat in other nostril, so all the medication is administered
- Continue ventilating patient as needed
- If seizures persist 5 minutes after treating, consider repeating ½ dose of midazolam either intranasally, intramuscularly or intravenously. Secure airway if necessary.
Midazolam Dosing chart
|
Patient age
(years) |
Weight
(kg) |
IN Midazolam volume in ml*
5mg/ml concentration Volume Dose (mg) |
|
Neonate |
3
kg |
0.3 ml
0.6 mg |
|
<1 yr |
6
kg |
0.4 ml
1.2 mg |
|
1 yr |
10 kg |
0.5ml
2.0 mg |
|
2 yr |
14 kg |
0.7 ml
2.8 mg |
|
3 yr |
16 kg |
0.8 ml
3.2 mg |
|
4 yr |
18 kg |
0.9 ml
3.6 mg |
|
5 yr |
20 kg |
1.0 ml
4.0 mg |
|
6 yr |
22 kg |
1.0 ml
4.4 mg |
|
7 yr |
24 kg |
1.1 ml
4.8 mg |
|
8 yr |
26 kg |
1.2 ml
5.2 mg |
|
9 yr |
28 kg |
1.3 ml
5.6 mg |
|
10 yr |
30 kg |
1.4 ml
6.0 mg |
|
11 yr |
32 kg |
1.4 ml
6.4 mg |
|
12 yr |
34 kg |
1.5 ml
6.8 mg |
|
Small teenager |
40 kg |
1.8 ml
8.0 mg |
|
Adult or full-grown teenager |
> 50 kg |
2.0 ml
10.0 mg |
* This volume is based on the calculated dose PLUS 0.10 ml dead space in the device (the amount of medication that will remain within the syringe and atomizer tip and therefore will not be delivered to the child). The total volume is then rounded off to the next highest 0.1 ml. Slightly higher doses may be appropriate at the lower range of volume (in smaller children) due to measurement difficulties and possible under dosing which may not stop the seizure.
In some children a higher dose (0.3 mg/kg) may be more appropriate
Teaching materials
Video of a family successfully treating their seizing child with intranasal midazolam in a city park
Home therapy for seizures:
- Download a condensed version of IN midazolam therapy for seizures as a PDF file
-
Download a basic photographic display of how to draw up and administer intranasal midazolam for seizures or sedation (click here for MS word document 0.53 MB)
- Down load file photo sequence of home IN therapy compiled by mother of epileptic child (Click here for PDF document 0.41 MB)
EMS seizure therapy protocols (Click here)
Peer Reviewed full articles open access from publisher
Lahat, IN midazolam for seizures, BMJ 2000 (click here) - PDF 0.25 MB
Krykou, IN midazolam for community seizure therapy, Epilepsy Australia 2006 (click here) - PDF 0.44 MB
Ma, Clinical Guidelines on Management of Prolonged Seizures, HK J Paediatrics 2010 - MS word filed
Segal, An alternate route of drug administration in acute convulsions, IMAJ 2000 (click here) - PDF 0.09 MB
Other articles and abstracts
- Whitesell, IN midazolam vs rectal diazepam - synthesis of the evidence, J Ped Healthcare 2009 abstract
- Ma, Clinical guidelines for status epilepsy in children, Hong Kong J Paediatrics 2010
- French JA.. Benzo vs Benzo: And the winner is.. Epilepsy Curr 2011;11(5):143-144.
Bibliography (click here for abstracts)
1.
DeLorenzo, R.J.,
et al., Epidemiology of status
epilepticus. J Clin Neurophysiol, 1995.
12(4): p. 316-25.
2.
Goodkin, H.P.,
J.L. Yeh, and J. Kapur, Status
epilepticus increases the intracellular accumulation of GABAA receptors.
J Neurosci, 2005. 25(23): p.
5511-20.
3.
Wermeling, D.P.,
et al., Bioavailability and
pharmacokinetics of lorazepam after intranasal, intravenous, and
intramuscular administration. J Clin Pharmacol, 2001.
41(11): p. 1225-31.
4.
Knoester, P.D., et
al., Pharmacokinetics and
pharmacodynamics of midazolam administered as a concentrated intranasal
spray. A study in healthy volunteers. Br J Clin Pharmacol, 2002.
53(5): p. 501-7.
5.
Malinovsky, J.M.,
et al., Plasma concentrations of
midazolam after i.v., nasal or rectal administration in children. Br
J Anaesth, 1993. 70(6): p.
617-20.
6.
Wermeling, et al.,
Pharmacokinetics and
pharmacodynamics of a new intranasal midazolam formulation in healthy
volunteers. Anesth Analg, 2006.
103(2): p. 344-9, table of
contents.
7.
Scott, R.C., F.M.
Besag, and B.G. Neville, Buccal
midazolam and rectal diazepam for treatment of prolonged seizures in
childhood and adolescence: a randomised trial. Lancet, 1999.
353(9153): p. 623-6.
8.
Camfield, P.R.,
Buccal midazolam and rectal
diazepam for treatment of prolonged seizures in childhood and
adolescence: a randomised trial. J Pediatr, 1999.
135(3): p. 398-9.
9.
Fisgin, T., et
al., Effects of intranasal
midazolam and rectal diazepam on acute convulsions in children:
prospective randomized study. J Child Neurol, 2002.
17(2): p. 123-6.
10.
Holsti, M., et
al., Prehospital intranasal
midazolam for the treatment of pediatric seizures. Pediatr Emerg
Care, 2007. 23(3): p. 148-53.
11.
Lahat, E., et al.,
Comparison of intranasal midazolam
with intravenous diazepam for treating febrile seizures in children:
prospective randomised study. Bmj, 2000.
321(7253): p. 83-6.
12.
Mahmoudian, T. and
M.M. Zadeh, Comparison of
intranasal midazolam with intravenous diazepam for treating acute
seizures in children. Epilepsy Behav, 2004.
5(2): p. 253-5.
13.
Wilson, M.T., S.
Macleod, and M.E. O'Regan,
Nasal/buccal midazolam use in the community. Arch Dis Child, 2004.
89(1): p. 50-1.
14.
Harbord, M.G., et
al., Use of intranasal midazolam
to treat acute seizures in paediatric community settings. J Paediatr
Child Health, 2004. 40(9-10):
p. 556-8.
15.
Jeannet, P.Y., et
al., Home and hospital treatment
of acute seizures in children with nasal midazolam. Eur J Paediatr
Neurol, 1999. 3(2): p. 73-7.
16.
Scheepers, M., et
al., Is intranasal midazolam an
effective rescue medication in adolescents and adults with severe
epilepsy? Seizure, 2000. 9(6):
p. 417-22.
18. Holsti, M, et al: Intranasal midazolam versus rectal diazepam for the home treatment of acute seizures in pediatric patients with epilepsy. Arch Pediatr Adolesc Med 2010;164(8):747-753. (click here for full article link)
19. McMullan J, Sasson C, Pancioli A, Silbergleit R. Midazolam versus diazepam for the treatment of status epilepticus in children and young adults: a meta-analysis. Acad Emerg Med 2010;17(6):575-82.
20. de Haan GJ, van der Geest P, Doelman G, Bertram E, Edelbroek P. A comparison of midazolam nasal spray and diazepam rectal solution for the residential treatment of seizure exacerbations. Epilepsia 2010;51(3):478-82.
21. Arya, R., S. Gulati, et al. (2011). "Intranasal versus intravenous lorazepam for control of acute seizures in children: A randomized open-label study." Epilepsia.
22. French JA.. Benzo vs Benzo: And the winner is.. Epilepsy Curr 2011;11(5):143-144 (click here for the open access article)
23. Anderson, M., et al., Pharmacokinetics of buccal and intranasal lorazepam in healthy adult volunteers. Eur J Clin Pharmacol, 2012. 68(2): p. 155-9.
24. Silbergleit, R., et al., Intramuscular versus intravenous therapy for prehospital status epilepticus. N Engl J Med, 2012. 366(7): p. 591-600.
25. Hirsch, L.J., Intramuscular
versus intravenous benzodiazepines for prehospital treatment of status
epilepticus. N Engl J Med, 2012.
366(7): p. 659-60.
26. Javadzadeh, M., K. Sheibani, et al. (2012). "Intranasal midazolam
compared with intravenous diazepam in patients suffering from acute
seizure: A randomized controlled trial." Iran J Pediatr
22(1): 1-8.
27. Hardmeier, M., R. Zimmermann, et al. (2012). "Intranasal
midazolam: pharmacokinetics and pharmacodynamics assessed by
quantitative EEG in healthy volunteers." Clin Pharmacol Ther
91(5): 856-862.
28.
Thakker, A. and P. Shanbag (2012). "A randomized controlled trial of
intranasal-midazolam versus intravenous-diazepam for acute childhood
seizures." J Neurol.
30. Humphries, L. K. and L. S. Eiland (2013). "Treatment of acute seizures:
is intranasal midazolam a viable option?" J Pediatr Pharmacol Ther
18(2): 79-87.
31. Sharma, R. and R. Harish (2013). "Comparative study of the efficacy of intranasal midazolam vs intravenous midazolam in convulsing neonates and children." RRJMHS 2(4): 54-57. (click for free article)
37. Inokuchi, R., N. Ohashi-Fukuda, et al. (2015). "Comparison of
intranasal and intravenous diazepam on status epilepticus in stroke
patients: a retrospective cohort study." Medicine (Baltimore)
94(7): e555.
39.
Kay, L., P. S. Reif, et al. (2015). "Intranasal midazolam during
presurgical epilepsy monitoring is well tolerated, delays seizure
recurrence, and protects from generalized tonic-clonic seizures."
Epilepsia.
40. Brigo, F., R. Nardone, et al. (2015). "Nonintravenous midazolam versus
intravenous or rectal diazepam for the treatment of early status
epilepticus: A systematic review with meta-analysis." Epilepsy Behav.
42. Bancke, L. L., H. A.
Dworak, et al. (2015). "Pharmacokinetics, pharmacodynamics, and safety
of USL261, a midazolam formulation optimized for intranasal delivery, in
a randomized study with healthy volunteers." Epilepsia
56(11): 1723-1731.
43.
Crawford, D. (2016). "Implementation of Intranasal Midazolam for
Prolonged Seizures in a Child Neurology Practice." J Neurosci Nurs.
44.
Haut, S. R., S. Seinfeld, et al. (2016). "Benzodiazepine use in seizure
emergencies: A systematic review." Epilepsy Behav
63: 109-117.
45.
Terry, D., A. D. Patel, et al. (2016). "Barriers to Seizure Management
in Schools: Perceptions of School Nurses." J Child Neurol.
47.
Alshehri, A., A. Abulaban, et al. (2017). "Intravenous Versus
Nonintravenous Benzodiazepines for the Cessation of Seizures: A
Systematic Review and Meta-analysis of Randomized Controlled Trials."
Acad Emerg Med 24(7):
875-883.
48.
Mula, M. (2017). "New Non-Intravenous Routes for Benzodiazepines in
Epilepsy: A Clinician Perspective." CNS Drugs
31(1): 11-17.
51.
Nunley, S., P. Glynn, et al. (2018). "Healthcare Utilization
Characteristics for Intranasal Midazolam Versus Rectal Diazepam." J
Child Neurol 33(2):
158-163.
52.
Ostendorf, A. P., K. Merison, et al. (2018). "Decreasing Seizure
Treatment Time Through Quality Improvement Reduces Critical Care
Utilization." Pediatr Neurol
85: 58-66.
53.
Kay, L., N. Merkel, et al. (2019). "Intranasal midazolam as first-line
inhospital treatment for status epilepticus: a pharmaco-EEG cohort
study." Ann Clin Transl Neurol
6(12): 2413-2425.
54.
Nunley, S., P. Glynn, et al. (2019). "A hospital-based study on
caregiver preferences on acute seizure rescue medications in pediatric
patients with epilepsy: Intranasal midazolam versus rectal diazepam."
Epilepsy Behav 92: 53-56.
55.
Owusu, K. A., M. B. Dhakar, et al. (2019). "Comparison of intranasal
midazolam versus intravenous lorazepam for seizure termination and
prevention of seizure clusters in the adult epilepsy monitoring unit."
Epilepsy Behav 98(Pt
A): 161-167.
56.
Theusinger, O. M., P. Schenk, et al. (2019). "Treatment of Seizures in
Children and Adults in the Emergency Medical System of the City of
Zurich, Switzerland - Midazolam vs. Diazepam - A Retrospective
Analysis." J Emerg Med 57(3):
345-353.
57.
Wallace, A., E. Wirrell, et al. (2019). "Seizure Rescue Medication Use
among US Pediatric Epilepsy Providers: A Survey of the Pediatric
Epilepsy Research Consortium." J Pediatr
212: 111-116.
58.
Detyniecki, K., P. J. Van Ess, et al. (2019). "Safety and efficacy of
midazolam nasal spray in the outpatient treatment of patients with
seizure clusters-a randomized, double-blind, placebo-controlled trial."
Epilepsia 60(9):
1797-1808.
 Therapeutic
Intranasal Drug Delivery
Therapeutic
Intranasal Drug Delivery