Intranasal Medications in Hospice and Palliative care:
This section of the web site will focus on data specifically related to intranasal drug delivery in patients with chronic pain being cared for by hospice or palliative care services. The primary issues discussed will be treatment of breakthrough pain, sedation of the acutely agitated patient and home therapy of seizures in the hospice setting. All three of these topics are covered in great detail for all patient populations in other sections of this web site and you are encouraged to visit the pain, sedation and seizure therapy sections of this web site by simply clicking on the appropriate tab in the upper left margin of this web page.
As of 2014 this topic has been overwhelmingly controlled by the pharmaceutical industry and does not require a site like this to bring it to light for others. Suffice to say - nasal opiates in hospice are now gold standard for breakthrough pain and there are multiple multidose, highly concentrated forms of fentanyl available to these patients.
Table of contents:
Treatment of breakthrough pain with transmucosal opiates - Introduction (click here)
Advantages of intranasal medications for breakthrough pain (click here)
Intranasal Opiates for breakthrough pain (click here)
Intranasal alfentanil (click here)
Intranasal Fentanyl (click here)
IN fentanyl palliative care literature (click here)
Intranasal sufentanil (click here)
Intranasal methadone (click here)
Intranasal morphine (click here)
Intranasal Ketamine in hospice and palliative care
What is ketamine? (click here)
Why use ketamine for pain? (click here)
Intranasal ketamine research in palliative care pain control (click here)
Intranasal ketamine and military research (click here)
Intranasal Medications for Home sedation and acute seizure control (click here)
Home Therapy of acute seizures (click here)
Treatment of breakthrough pain: Introduction
Breakthrough pain is a transient flare-up of moderate to severe pain in patients with chronic pain conditions who are otherwise well controlled with a stable analgesic regimen. The problem is not minor – in the United states alone it is reported than 800,000 cancer patients suffer from breakthrough pain.[1, 2]
Many treatments are proposed for treating breakthrough pain, including numerous protocols on the internet, however few randomized controlled trials (high level of evidence) comparing pain medication treatment options are currently available.[2] Interestingly, the majority of the randomized controlled trials that do exist regarding drug therapy for breakthrough pain have investigated transmucosal drug delivery options such as nasal, sublingual and buccal drug delivery.[2-14] The reason for this is probably related to the unique advantages that transmucosal drugs offer. Because breakthrough pain is by definition rapid in onset and brief in duration, the ideal characteristics of a breakthrough pain medication will be rapid effect, early peak and short duration of action with minimal side effects or sedation.[1] Transmucosal drug delivery - especially via the nasal mucosa - offers all of the above mentioned features of an “ideal” medication for breakthrough pain. Nasal transmucosal pain medications exert clinical effect within 5 minutes, peak at 10 to 30 minutes and wear off in 1-2 hours. Furthermore, if packaged properly they are easily administered by patients, family or caregivers, they are painless to administer (no shots), the area of administration (nose) is easily accessed, they offer unique CNS bioavailability due to the nose-brain pathway, they can be administered even in patients who are nauseated or having difficulty swallowing and if highly concentrated generic solutions are obtained they are relatively inexpensive.[15]
Advantages of Intranasal drug delivery for breakthrough pain:
| Ideal BTP drug features | I.N. | I.V. | S.C. I.M. |
S.L. | P.O. | P.R. |
| Rapid onset | Yes | Yes | +/- | +/- | No | No |
| Early Peak | Yes | Yes | +/- | +/- | No | No |
| Short duration | Yes | Yes | +/- | Yes | No | No |
| Painless administration | Yes | No | No | Yes | Yes | +/- |
| Easy delivery by patient | Yes | No | No | Yes | Yes | No |
| Easily delivered to non-cooperative patient | Yes | Yes | No | No | No | +/- |
| Not effected by mucositis | Yes | Yes | Yes | No | No | Yes |
| Rapid transport to CNS | Yes | Yes | No | No | No | No |
The caveat here in terms of successful I.N. delivery is that the drugs need to be both highly concentrated and highly lipophilic (easily cross cellular membranes). Fortunately multiple inexpensive IV analgesic drugs fit this description and can be reliably delivered via the nasal mucosa: Fentanyl, sufentanil, methadone and ketamine.
Intranasal Fentanyl, alfentanil and Sufentanil for hospice and palliative care:
The synthetic fentanyl based opiates alfentanil, fentanyl and sufentanil are all highly lipophilic and appear to be ideal opiates for transmucosal delivery with measurable plasma levels within seconds of exposure to the nasal mucosa.[16] They are far more bioavailable across the oral and nasal mucosa than morphine and they are far more potent allowing small volumes to be administered effectively.
| Alfentanil | Fentanyl | Sufentanil | |
| Potency compared to morphine | About 30:1 | 100:1 | 600 to 1000:1 |
| Onset of action | 3-5 minutes? | 5 minutes | 2-5 minutes |
| Half life | 1.5 hours | 3 hours | 2.6 hours |
| Duration of pain control (I.N.) | Unclear - 10 minutes to 1 hour? | 1 hour | 1 hour |
| Generic concentration | 500 mcg/ml | 50 mcg/ml | 50 mcg/ml |
| "Ideal" concentration for intranasal delivery if compounded | Unclear. Probably works in current concentration but would be better if doubled | 500 mcg/ml | Current generic concentration is ideal for I.N. delivery |
| Reasonable I.N. starting dose - titrate to effect with dosing trial | 5 mcg/kg? | 1.5 to 2 mcg/kg | 0.4 to 0.5 mcg/kg |
Intranasal Alfentanil:
Alfentanil has the least number of published articles confirming its efficacy in any clinical setting, though its use is described in the hospice setting by practitioners in England.[17, 18] A disadvantage of this medication compared to the other synthetic opiates for nasal drug delivery is the lower potency. However this appears to be overcome by high concentration of the drug. An advantage and potential disadvantage of Alfentanil is its very short duration of action – it been reported to begin wearing off within 10 minutes in discussions from palliative services in England. This may be ideal for very acute and short lasting painful spells, but could be too short a duration for longer spells. Until more literature is published on intranasal Alfentanil it is unclear if concentration and duration of action are issues or not.
Osborn et al: Intranasal alfentanil for severe intractable angina in inoperable coronary artery disease. Palliative Medicine 2009[18]
This article is a case report describing a man who had very difficult to treat anginal pain leading to recurrent hospital admissions. Palliative care was involved and they found that 280 mcg (two nasal sprays of 140 mcg per spray of alfentanil 1mg/ml – or 1000 mcg/ml) of alfentanil relieved his anginal pain within 5 minutes. At the time of the report he has successfully used this therapy 4-6 times per week for over 6 months with continued good effect.
Other information on clinical efficacy of intranasal Alfentanil:
St. Christopher’s Hospital in London England touts the application of nasal alfentanil delivered via a multidose pump atomizer as a highly effective method for treating acute breakthrough pain spells. They use alfentanil in 1 mg/ml concentrations and titrate to effect – reporting onset in 3 minutes and a fairly brief duration of action lasting approximately10 minutes. You can read about their experience on-line at www.palliativedrugs.com
Intranasal Fentanyl:
Intranasal fentanyl, a drug that is 100 times as potent as morphine is the most extensively studied synthetic opiate for treating pain either in the hospice setting or in other painful situations (see pain section of this web site).[19] A comprehensive review of transmucosal fentanyl was posted on www.palliativedrugs.com in August of 2009 and readers with interest should visit and download this document (090916_Fentanyl August 2009_2).[20] It describes the literature to date regarding oral transmucosal fentanyl, sublingual fentanyl and intranasal fentanyl in the setting of chronic pain. The document points out that intranasal fentanyl is superior to oral transmucosal fentanyl, which in turn is superior to oral morphine for speed of onset and efficacy in reducing the intensity of break through pain:
“All fentanyl treatments were more effective than placebo in treating breakthrough pain at all time points. OTFC was more effective than MSIR in treating breakthrough pain at all time points. Intranasal fentanyl provided a greater reduction in pain at 10 minutes compared to that provided by the buccal tablets and oral mucosal lozenges at 15 minutes (see table 2). The superior pain reduction achieved with the intranasal spray compared with the other interventions was maintained for up to 45 minutes after administration.”[20]
Table 2: Treatment effects (pain intensity difference) relative to placebo[20]
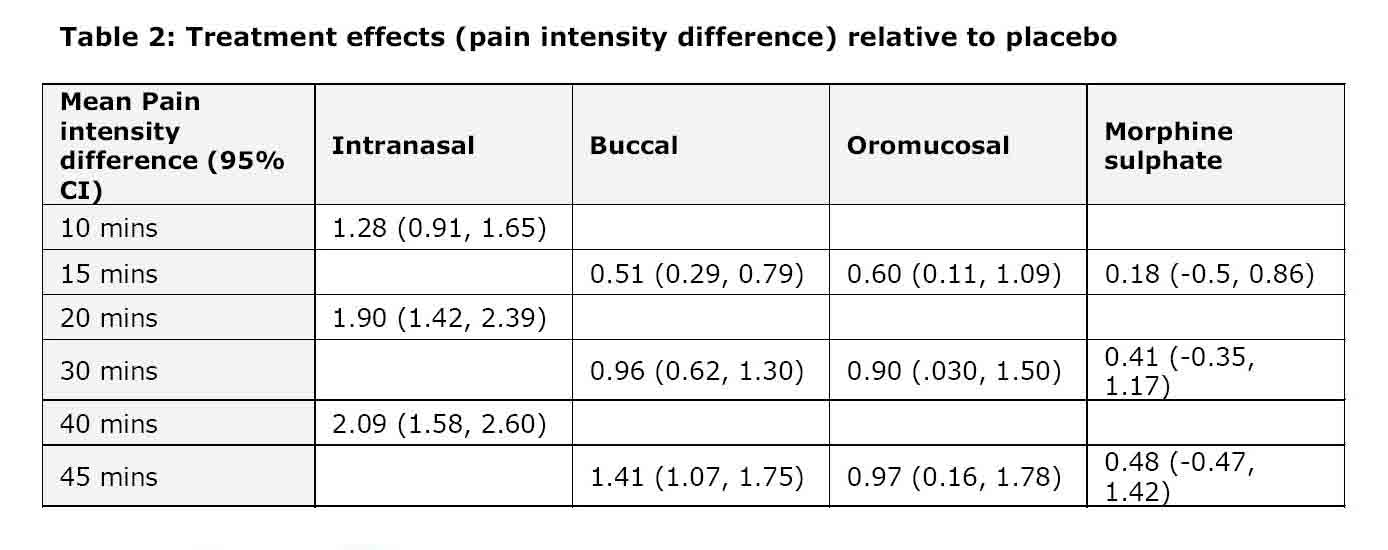
The Author also provides clinical data regarding drug bioavailability and onset of action:
| Bioavailability | Onset of pain relief | Peak Pain Relief | Duration of action | |
| Sublingual Fentanyl | 70% | 15 min | 22-240 min | Hours |
| Oral Transmucosal Fentanyl lollipops | 25% via buccal 75% via gut |
20 min | 20-40 min | Hours |
| Rapid absorbing OTMC fentanyl on tongue | 65% (half via buccal route) | 15 min | 47 min | Hours |
| Intranasal Fentanyl | 89% | 5 min | 12-15 min | 1 hour |
The document further suggests that dosing decisions for intranasal drugs are not reliably determined using the traditional 1/6 the daily dose equivalency. (A comment verified by subsequent research by Mercadante and Good in articles reviewed below).[1, 12] Instead, a new dosing titration phase needs to be conducted – starting with a relatively low intranasal dose and titrating to effect until the appropriate dose is determined. Finally the document reiterates that intranasal fentanyl has an onset of action within 5 minutes and peaks in 10 to 20 minutes with a duration of approximately 1 hour – a time line that is appropriate for breakthrough pain. Finally the document presents cost data for oral transmucosal drugs based on charges in England in British pound sterling – demonstrating the fairly expensive individual doses of buccal and sublingual brand name products (see below). Generic intranasal drugs will likely cost less even if they need to be compounded to adequate concentrations (fentanyl) for nasal delivery (sufentanil does not need compounding – its generic formulation is ideal for nasal delivery).
Other authors also note that IN fentanyl provides reliably faster pain relief compared to oral transmucosal fentanyl (see Caplan Meier curves below).[53]
Critical concept: Drug concentration and volume of administration
Once again - as stated throughout this web site – dosing volumes are critical. Generic fentanyl is probably too dilute for reliable and reproducible treatment of adult patients in severe pain because the generic formulation is 50 mcg/ml and a typical dose for a patient with severe pain is 1 to 2 mcg/kg – meaning a 70 kg patient would need 1.4 to 2.5 ml of drug in the nose to achieve the 70 to 140 mcg dose. For this reason the recently approved intranasal fentanyl (Instanyl) comes in concentrations of at least 500 mcg/ml, allowing very small volume dosing to treat acute breakthrough pain spells.[12] If Instanyl is not on your formulary (it is not yet available in the USA) or if it becomes cost prohibitive there are two inexpensive solutions: Either fentanyl compounded by your pharmacy to a similar concentration, or generic sufentanil which also comes in 50 mcg/ml solution but because it is 10 times as potent as fentanyl – it has equivalency to the 500 mcg/ml fentanyl solution.
Intranasal fentanyl for palliative care - published literature:
Mercadante, S., et al., A comparison of intranasal fentanyl spray with oral transmucosal fentanyl citrate for the treatment of breakthrough cancer pain: an open-label, randomised, crossover trial. Curr Med Res Opin, 2009[12]
This is a landmark trial regarding intranasal fentanyl for the treatment of breakthrough pain. The authors randomized 139 patients to either oral transmucosal fentanyl – OTFC (Actiq) versus intranasal fentanyl (Instanyl) in a cross over trial where the patients served as their own control by being subjected to both treatments. Instanyl is a highly concentrated form of fentanyl – ranging from 500 mcg/ml to 2000 mcg/ml delivered at a 0.1 ml dose of 50 to 200 mcg) into one nostril, with the option of a second dose at 10 minutes via an atomizer device. Patients had to have cancer associated breakthrough pain occurring at least 3 times per week to be eligible. In the initial part of the trial they had a dose titration phase to determine both the proper dose of Actiq and of Instanyl to control their usual pain – this dose was then used for treatment of breakthrough pain during the randomization phase. Once dosing was determined patients were treated with either method for a maximum of 2 weeks until they had used the study drug 6 times, at which point they crossed over and used the alternate drug six times. The patients were not blinded to the treatment medication. The primary outcome was time until meaningful pain reduction as per patients opinion. Secondary outcome was pain relief based on a numerical rating scale (0 to 10) with time to achieving at least 33% reduction and 50% reduction recorded.
Picture of Instanyl:
Results: Intranasal fentanyl resulted in a mean time to meaningful pain reduction of 11 minutes whereas OTFC onset of meaningful relief was 16 minutes. At 5 minutes 25% of patients had > 33% pain reduction using nasal fentanyl compared to only 7% of OTFC. At 10 minutes these percentages had increased to 51% and 24% respectively. By 20 to 30 minutes 80-90% of patients had at least 33% pain reduction using nasal fentanyl compared to 60-80% using OTFC. The table below shows the results of pain relief both at 33% reduction and 50% reduction.
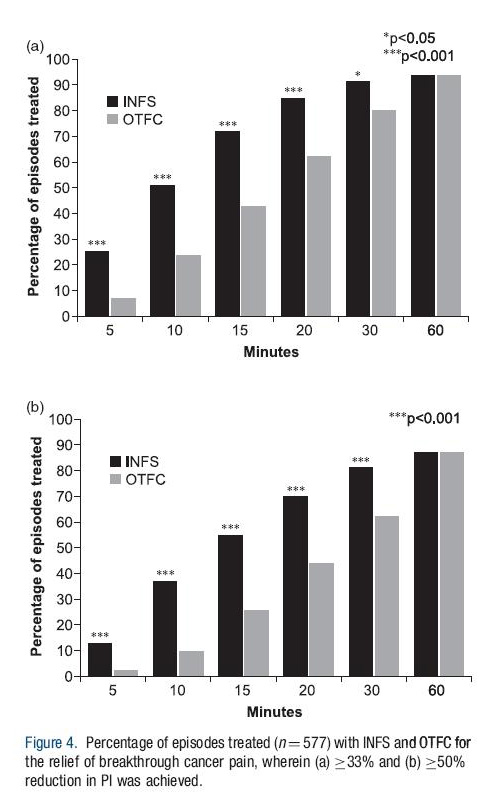
Although both drug deliver methods were well tolerated, the patients (77%) strongly preferred intranasal fentanyl compared to OTFC delivery. The authors conclude that intranasal fentanyl resulted in significantly faster onset of meaningful relief of cancer associated breakthrough pain and that it was preferred to oral transmucosal fentanyl for this indication.
Review article regarding Instanyl for breakthrough pain: Chaplin 2010
Kress, H.G., et al., Efficacy and tolerability of intranasal fentanyl spray 50 to 200 microg for breakthrough pain in patients with cancer: a phase III, multinational, randomized, double-blind, placebo-controlled, crossover trial with a 10-month, open-label extension treatment period. Clin Ther, 2009[4]
In this study 111 patients were randomized to to either intranasal placebo or intranasal fentanyl in a cross over trial where the patients served as their own control by being subjected to either treatment during 8 separate episodes of breakthrough pain over a 3 week time period. The patients were asked to use the study drug, then could use their standard formulation of pain control for breakthrough pain if they had inadequate pain control by 10 minutes. Following the blinded study arm, they were allowed to continue open label use of the nasal fentanyl for up to 10 months in a tolerability study. The intranasal fentanyl formulation used is a highly concentrated form of fentanyl – ranging from 500 mcg/ml to 2000 mcg/ml delivered at a 0.1 ml dose of 50 to 200 mcg) into one nostril, with the option of a second dose at 10 minutes via an atomizer device. In the initial part of the trial they had a dose titration phase to determine both the proper dose of intranasal fentanyl to control their usual pain – this dose was then used for treatment of breakthrough pain during the randomization phase. The primary outcome was the difference in pain scores by 10 minutes after application of the nasal drug vs placebo.
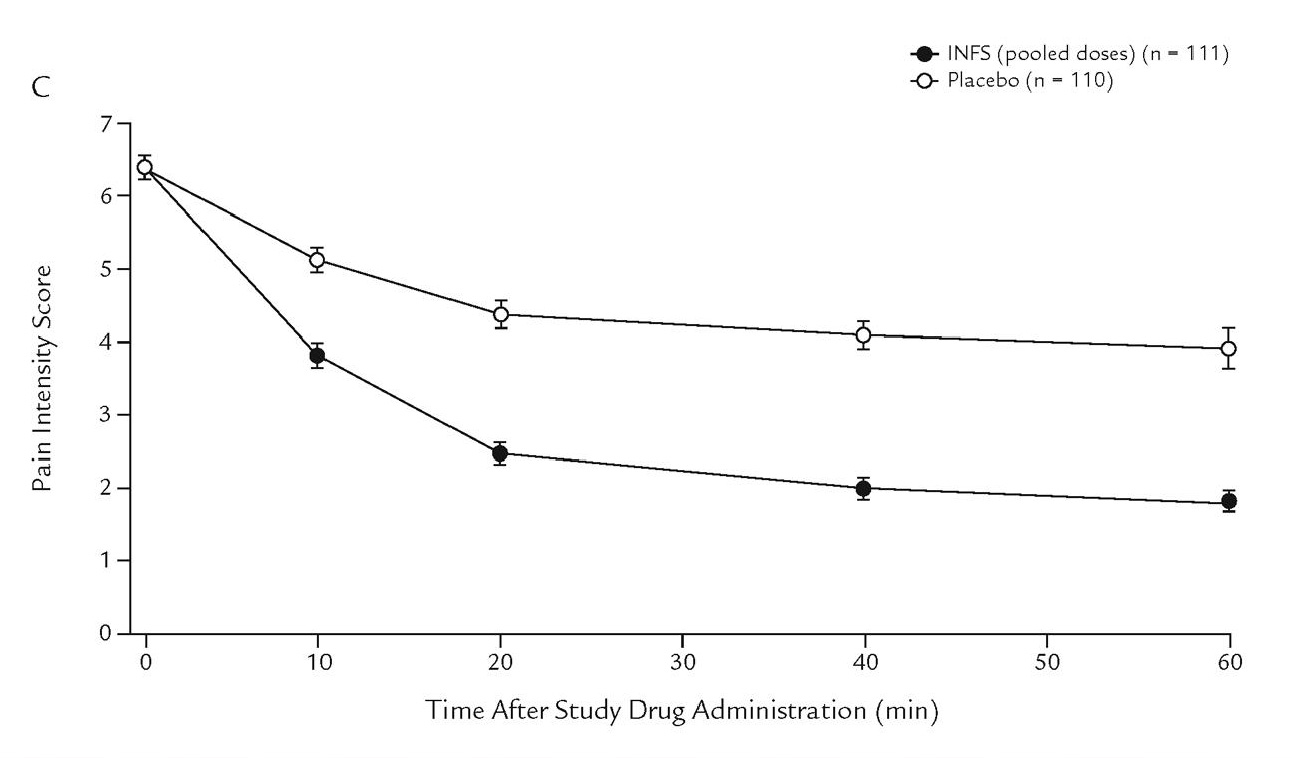
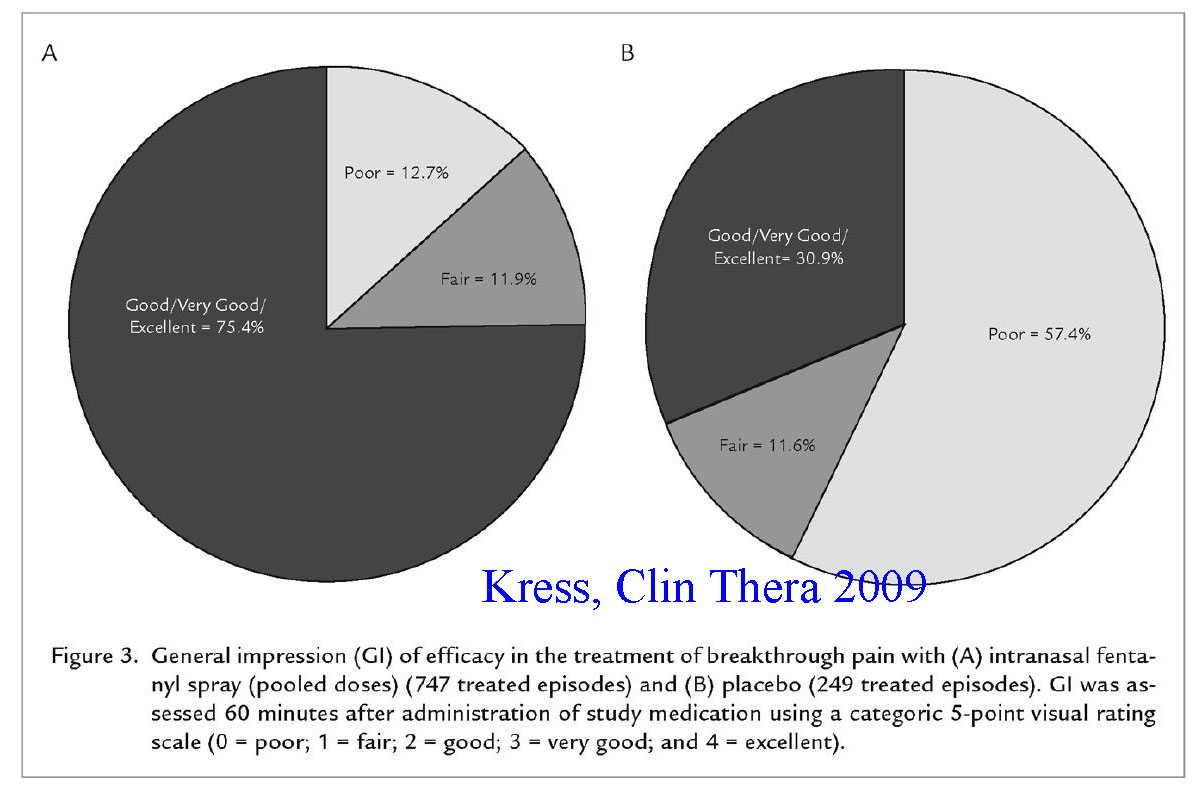
The results showed a significant drop in pain intensity scores (twice that of placebo) by 5 minutes that persisted for the entire 60 minute study. At least a 33% reduction in pain had occurred in 57% of IN fentanyl patients vs 28% of placebo cases by 10 minutes. Rescue doses of primary medication were used by 14% of intranasal fentanyl patients compared to 45% of placebo patients. The drug was well tolerated during the extension treatment period with most withdrawals from use related to cancer progression.
Zeppetella, G., An assessment of the safety, efficacy, and acceptability of intranasal fentanyl citrate in the management of cancer-related breakthrough pain. A pilot study. J Pain Symptom Manage, 2000[21]
This study is one of the first to investigate intranasal fentanyl for the treatment of cancer related breakthrough pain (BTP). In an open label fashion they allowed 12 patients to administer a total of 20 mcg of fentanyl to the nose for acute treatment of BTP. They noted 66% of patients had pain relief by 10 minutes, but only half of the group of patients had good to excellent pain relief. While this study confirmed that IN fentanyl could work, it has some significant design flaw which include a dose that is far to low to be expected to work (20 mcg is not even an IV dose) and they used fentanyl at concentrations of only 50 mcg/ml which are likely too dilute for adequate dosing of 1 to 2 mcg/kg initial dose. Never the less this research was an important first step to assist with future study designs.
Zeppetella, G., Nebulized and intranasal fentanyl in the management of cancer-related breakthrough pain. Palliat Med, 2000.[22]
Nebulized fentanyl is being developed for treatment of BTP, however it remains to be determined whether it is safe to deliver drugs to the lung and not cause damage and whether there is any advantage over more simple delivery to the nose.
Sitte, T. and C. Bausewein, Intranasal fentanyl for episodic breathlessness. J Pain Symptom Manage, 2008[23]
Dr. Sitte describes three patients with severe episodic breathlessness that he successfully and rapidly treated with intranasal fentanyl (which he has his pharmacy compound to high concentrations. He further states that now after over 200 patients in whom he has treated at home for dyspnea, this is a safe and effective therapy and challenges someone to conduct a trial to support his findings.
Taylor, et al: Pharmacokinetic and local tolerability profiles of three potential fentanyl nasal spray formulations developed for breakthrough cancer pain compared with oral transmucosal fentanyl citrate. J Clinical Oncology 2009 Abstract (Vol 27, No15S May)[52]
These authors compared IN fentanyl (3 formulations linked to a mucosal adhesive) to OTMF and measured plasma venous concentration and bioavailability. They found peak plasma levels to be far earlier with nasal formulations (10 to 20 minutes) compared to OTMF (90 minutes). they also noted far higher bioavailabilities. The authors conclude "Compared with OTFC, all three fentanyl nasal spray formulations demonstrated enhanced pharmacokinetic profiles appropriate for breakthrough cancer pain as evidenced by significantly increased systemic exposure and reduced times to peak plasma values."
Davis, Rapid pain relief with fentanyl citrate nasal spray in cancer patients with breakthrough pain, EAPC 2007
This poster presentation at an international pain conference in 2007 showed the results of a small study using intranasal fentanyl to treat breakthrough pain. By five minutes about ¼ of patients were experiencing meaningful pain relief while 60 percent of patients has meaningful pain relief by 10 minutes and 86% by 20 minutes.
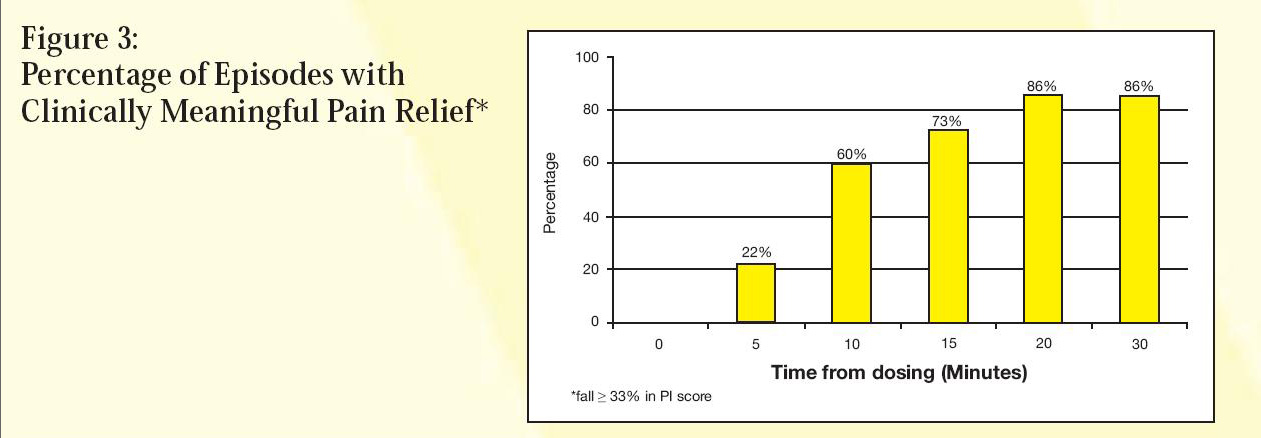
Click here to download the entire poster presentation by Davis et al
Harlos, M., Palliative Care Incident Pain and Incident Dyspnea Protocol. Internet public domain protocol, 2002: p. http://palliative.info/incidentpain.htm.[24]
This web site provides a treatment protocol using generic intranasal fentanyl (or sufentanil) for both breakthrough pain and or dyspnea based on their extensive personal experience.
Intranasal Sufentanil:
Sufentanil is in the same family as fentanyl, is also generic, but is 10 times as potent as fentanyl. For this reason smaller volumes/doses can be used with similar effect as a higher fentanyl dose. This drug is so lipophilic that as soon as it touches the mucosal membrane of the nose, detectable levels are measurable in the blood stream.[16] Given its high bioavailability, rapid onset of action with relatively short duration, high therapeutic index and the fact that in currently packaged generic concentrations/volumes it works extremely well for intranasal administration this drug is arguably the ideal medication for cost effective treatment of breakthrough pain. The caveat is that dose titration is required to determine effective and safe doses since high doses can lead to respiratory depression (editor personal experience).
Intranasal sufentanil for palliative care - published literature:
Good, P., et al., Intranasal sufentanil for cancer-associated breakthrough pain. Palliat Med, 2009[1]
Good et al studied intranasal sufentanil effectiveness for treating breakthrough pain in 30 cancer patients. The investigators first step was to determine the effective and safe dose of nasal sufentanil by incrementally increasing the dose administered until an effective but non-sedation dose was determined (dose titration phase). They administered the drug with a bottle atomizer delivering about 9 mcg per spray. They found the mean effective dose to be 18 mcg with a range of 9 to 108 micrograms per dose. No significant adverse effects (over sedation or respiratory depression) occurred during dosing leading the authors to suggest they were perhaps overly cautious during the titration phase and allowing them to be very comfortable with this drug being used in the home setting. They then sent the patients home with the delivery device and allowed them to administer intranasal sufentanil as needed for breakthrough pain. The bottom line – 77% found it as effective as prior medications and 94% preferred it as their first line treatment for breakthrough pain. The authors conclude that intranasal sufentanil “provides acceptable and often preferred breakthrough analgesia for many patients and most importantly, it can provide rapid pain relief. This study also showed that IN sufentanil can be used safely, with a very low incidence of adverse effects, in an inpatient palliative care population. We suggest that IN sufentanil be added to the armamentarium of medication used to treat breakthrough pain with the proviso that it requires an initial dose titration phase.”

The above graph demonstrates drop in verbal rating scores for breakthrough pain following use of intranasal sufentanil
Jackson, K., M. Ashby, and J. Keech, Pilot dose finding study of intranasal sufentanil for breakthrough and incident cancer-associated pain. J Pain Symptom Manage, 2002. 23(6): p. 450-2.[25]
These authors are some of the same authors as the above Good trial and it may be that the 4 patients and 7 episodes of treatment described are included in the Good trial. Bottom line is that they describe good pain control in 5 of 7 episodes with relatively low doses of sufentanil – and that the pain control lasted up to 2 hours.
Gardner-Nix, J., Oral transmucosal fentanyl and sufentanil for incident pain. J Pain Symptom Manage, 2001[26]
This study describes 6 cases – 3 who were treated with transmucosal sufentanil with good results. It was one of the original papers describing sufentanil for incident pain, but does not really quantify effect.
Harlos, M., Palliative Care Incident Pain and Incident Dyspnea Protocol. Internet public domain protocol, 2002: p. http://palliative.info/incidentpain.htm.[24]
This web site provides a treatment protocol using generic intranasal fentanyl (or sufentanil) for both breakthrough pain and or dyspnea based on their extensive personal experience.
Intranasal Methadone for hospice and palliative care:
Due to its high lipophilicity, methadone should easily crosses the nasal mucosal membrane. However, we are unable to locate any literature on this topic and are only aware of anecdotal discussions. Further research into this concept may be appropriate, though the prolonged length of action of methadone probably makes it a poor candidate for brief treatment of breakthrough pain.
Intranasal morphine for hospice and palliative care:
Unfortunately, morphine – our most inexpensive opiate – is not as readily available across the mucous membranes as some of the other opiates due to its non-lipophilic molecular structure. In bioavailability studies only about 10% of the drug crosses the nasal mucosa, the remainder either not absorbing or being swallowed and undergoing hepatic first pass metabolism prior to being available long after administration. For these reasons – even though commonly prescribed as a sublingual and buccal drug – transmucosal morphine often leads to slow and inadequate pain relief for acute breakthrough pain spells.
Morphine that is modified with adjuvants that assist nasal adherence and absorption (such as Morphine-6- glucuronide and morphine with chitosan) is much more bioavailable across the nasal mucosa but is currently not available for clinical use though that may soon change.[27] Diamorphine (pharmaceutical heroin) is highly bioavailable and comes packaged as a lyophilized powder allowing reconstitution in very high potencies – minimally diluted to the appropriate volume for any size patient such that only a small volume (0.2 ml) needs to be administered to the mucosa.[28, 29] Once in the blood stream diamorphine is converted to morphine and it metabolites where it exerts its clinical effect. This medication is commonly prescribed in United Kingdom emergency departments for treating acute fractures in children and there is some discussion of its use at the end of life on palliative care web sites.[28] However, diamorphine is not readily available outside the United Kingdom so is not really an option in other locations. Fortunately the synthetic opiates of the fentanyl family are readily available in most countries, are generic and are highly bioavailable across the nasal mucosa.
Intranasal ketamine for hospice and palliative care:
What is ketamine?
Ketamine is dissociative anesthetic agent that has analgesic properties when used in sub-anesthetic doses. Its site of action is the dorsal horn of the spinal cord where it blocks N-methyl D-aspartate (NMDA) receptors – hence the term NMDA antagonist. Because it has a different site of action than opiates, it is often useful as an alternate therapy painful conditions that are poorly controlled by first line analgesics. It is especially useful for neuropathic pain but also in situations such as phantom limb pain, ischemic pain and incident pain such as during dressing changes and movement. It suffers from a number of side effects that are somewhat dose related (therefor less common in analgesic doses as compared to anesthetic doses): Dysphoria, vivid dreams/nightmares, hallucinations, feelings of unreality, diplopia, nystagmus, hypertension and tachycardia. Furthermore, though ketamine itself does not cause respiratory depression, there is some evidence that it potentiates the action of opiates and this might cause some increased sedation or respiratory depression in patients on very high doses of opiates. Due to the known side effects, conservative recommendations suggest that ketamine should be avoided in patients with psychotic disorders, arrhythmias, severe hypertension or elevated intracranial or intraocular pressure.
Click here for a slide set 2010 review of ketamine
Why use ketamine for breakthrough pain?
Ketamine is labeled as a general anesthetic drug and in anesthetic doses causes a dissociated catatonic state where patients stare into space – seemingly awake, breathing – but they do not react to painful stimuli. In these doses it can be used for very painful procedures such as joint and bone reduction and other orthopedic and plastic surgical interventions. However, ketamine is also an analgesic drug and demonstrates analgesic properties at doses 10-15 times less that that required for anesthesia. Because of this analgesic effect at low doses, patients often remain completely awake and alert while obtaining substantial relief of their pain. Numerous studies now confirm the analgesic effect of ketamine in low doses when used to treat neuropathic pain, phantom limb pain, post-operative and traumatic pain as well as during dressing changes.[30-33]
The combination of efficacy for treating neuropathic and incident pain, alternate mechanism of action from opiates, minimal side effects at analgesic dose ranges, inexpensive cost, high concentration and lipophilicity has led investigators to study this medication in an intranasal form as a potential treatment option for breakthrough pain in the hospice and palliative care setting.
Intranasal ketamine research for hospice and palliative care:
Please see the PAIN section and / or the SEDATION section of this web site for discussion of intranasal ketamine in the non-hospice setting
Kulbe J, The use of ketamine nasal spray for short-term analgesia. Home Healthcare Nurse 1998[33]
This is a single patient case study describing a 96 year old woman with burns that needed daily dressing changes done at home. She had very poor pain control with 1st line analgesics – failing to obtain adequate pain control for dressing changes with oxycodone and acetaminophen. After introducing intranasal ketamine – 15 mg total dose - she tolerated the dressing changes very well with no significant side effects. This simply suggests that in the properly selected individual, intranasal ketamine is an option worth considering when other options are inadequate
Carr et al, Safety and efficacy of intranasal ketamine for the treatment of breakthrough pain in patients with chronic pain: a randomized, double-blind, placebo controlled, crossover study. Pain 2004[34]
Dr. Carr and colleagues conducted a dosing and efficacy trial comparing intranasal ketamine to placebo in 20 patients. Their study was a well-designed double blind randomized controlled trial with patients as their own crossover. In this trial, patients with chronic pain and at least 2 episodes of breakthrough pain per day were randomized to nasal ketamine vs nasal placebo if their pain scores were 5 or greater. They were allowed to administer 1 atomized spray of ketamine or placebo every 90 seconds into alternate nostrils until pain was improved to a maximum of 5 doses in 7.5 minutes (0.1 ml per spray = 10 mg ketamine or 50 mg maximum if 5 sprays were administered. The ketamine was a 10% solution – 100 mg/ml of off the shelf IV formulated ketamine). The delivery device was a 20 ml amber bottle prefilled with the drug or placebo that had a positive displacement pump screwed onto the top and had a nasal atomizer tip as the delivery mechanism. It was actuated by manual compression by the patient. The results were fairly impressive – mean pain scores dropped by 2.65 units with ketamine vs 0.81 with placebo. 65% of patients using ketamine had at lead a 40% reduction in their pain scores, while only 20% of placebo patients achieved similar reductions in pain.
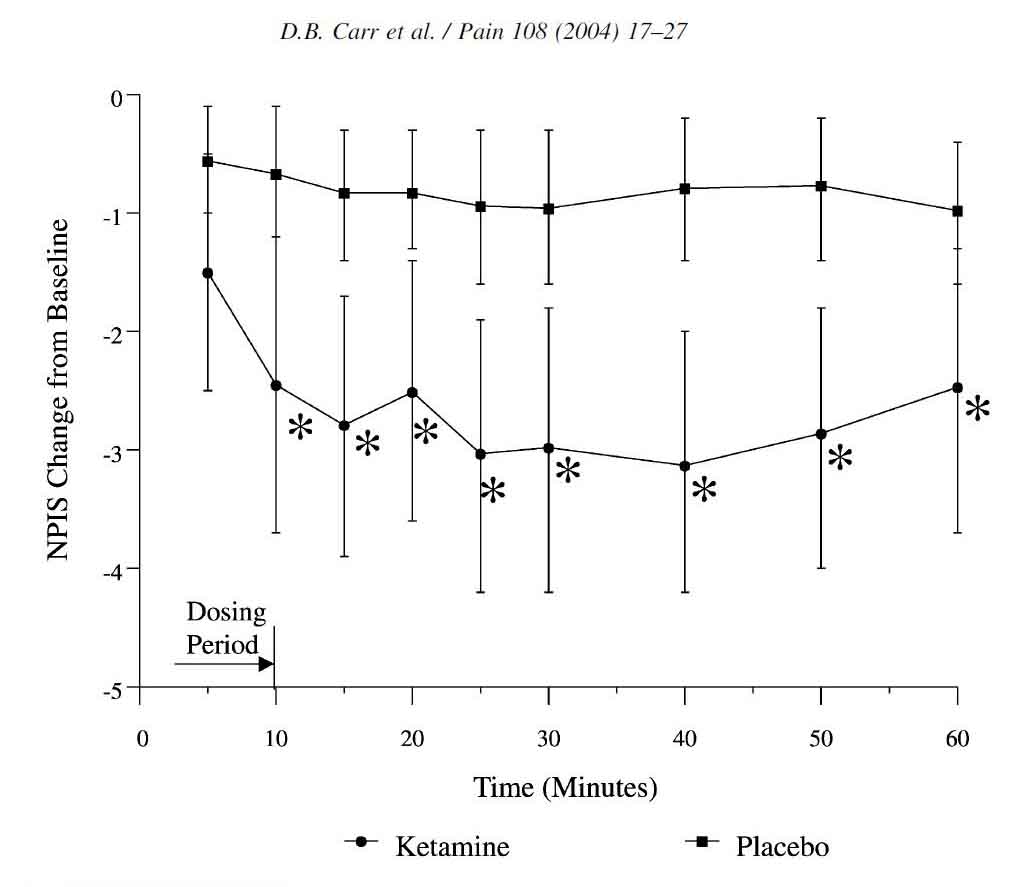
Pain relief occurred within 10 minutes and no patients needed their rescue medications in the IN ketamine group. Side effects were minimal and no patient reported visual or hallucinatory type events. The authors call for larger trials of this unique and promising treatment for break through pain. This study demonstrates that adequately concentrated ketamine (100 mg/ml) at relatively low doses and low volumes (10 to 50 mg, or 0.1 to 0.5 ml total) is effective for breakthrough pain. [Web site Editor note - Future researchers should be careful to mimic these volumes (0.5 ml or less per nostril if at all possible) as ketamine comes in many concentrations and lower concentration may result in unacceptable volumes delivered to the nose with an expected less than adequate outcome. In fact – ketamine is available as 15% solution (15 mg/ml) which might be even more appropriate.]
Huge, Effects of low-dose intranasal (S)-ketamine in patients with neuropathic pain. Pain 2009[35]
Dr. Huge et al conducted a randomized trail comparing two separate doses of intranasal racemic s-ketamine (Ketanest) to determine the effective dose for treating neuropathic pain. In this study patients with neuropathic pain were randomized to either 0.2 mg/kg or 0.4 mg/kg of ketamine. Unfortunately they used a concentration of only 25 mg/ml and further diluted it to 3 ml total volume regardless of the patients weight. Despite this dilution they still demonstrated a reduction in pain scores from baseline of 6.4 to 6.6 down to 4.4 and 4.1 respectively for the two concentrations (0.2 mg/kg, 0.4 mg/kg – 30 to 40% drops in pain scores).
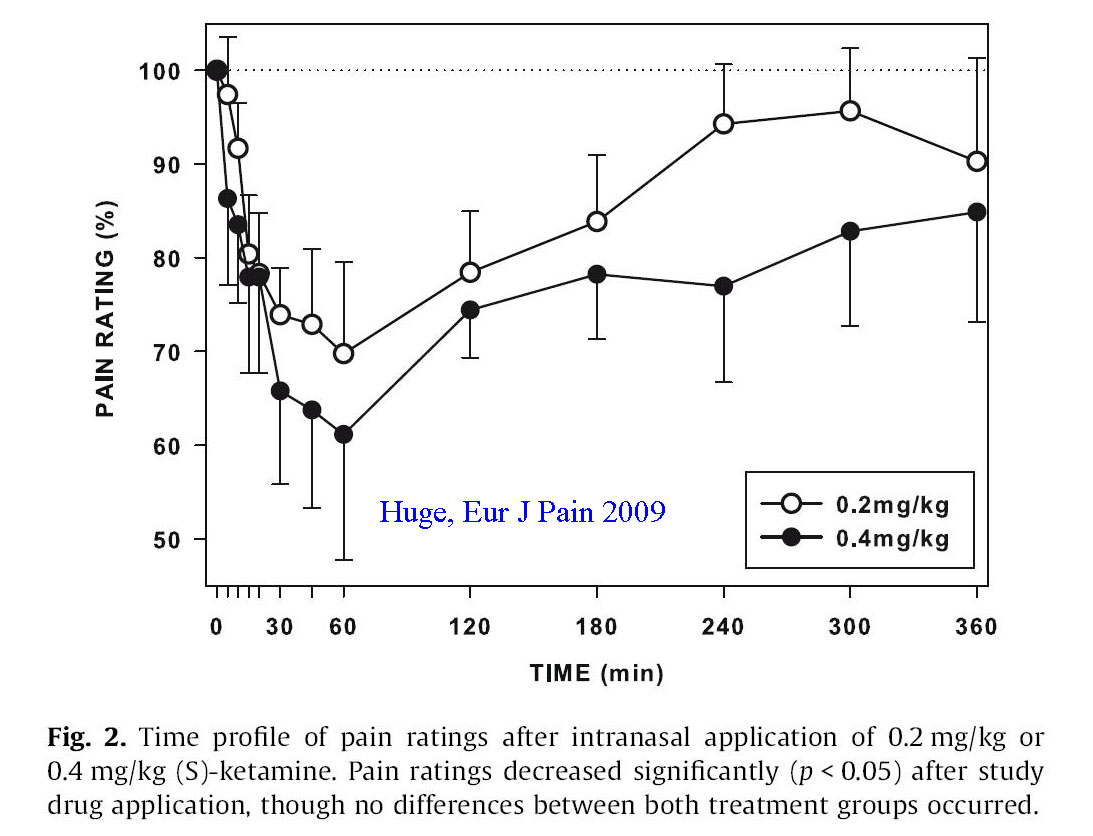
The authors conclude that intranasal ketamine at low doses shows promise for treating neuropathic pain, but admit that volumes of 3 ml are impractical and likely results in substantial swallowing and first pass metabolism by the liver. They recommend lower volumes of more potent ketamine to enhance nasal mucosal delivery and reduce oral delivery.
United States Military data regarding intranasal ketamine for incident pain
The U.S. military may be the group most interested in intranasal ketamine for treating “incident” pain. This group is looking for a pain medication that can be self administered and relieve a soldiers acute pain related to war injury, yet let him/her still function at a high level and either be able to extricate himself or finish the mission (often saving his/her life and others by being a functional asset rather than a detriment). To date they have funded and conducted numerous studies (including the Carr study above) to find the right dose of IN intranasal ketamine while determining the incidence of side effects. Looking at the material posted on the internet, they have studied at least 117 subjects with doses ranging from 10 to 50 mg of ketamine finding no serious effects and adequate pain control. The graph below notes their results in terms of dosing equivalency – finding 50 mg of nasal ketamine as effective or more effective than 7.5 mg of IV morphine without the loss of function seen with morphine. Look for more literature on this topic in the near future. (click here for 2005 report from the military on this topic)
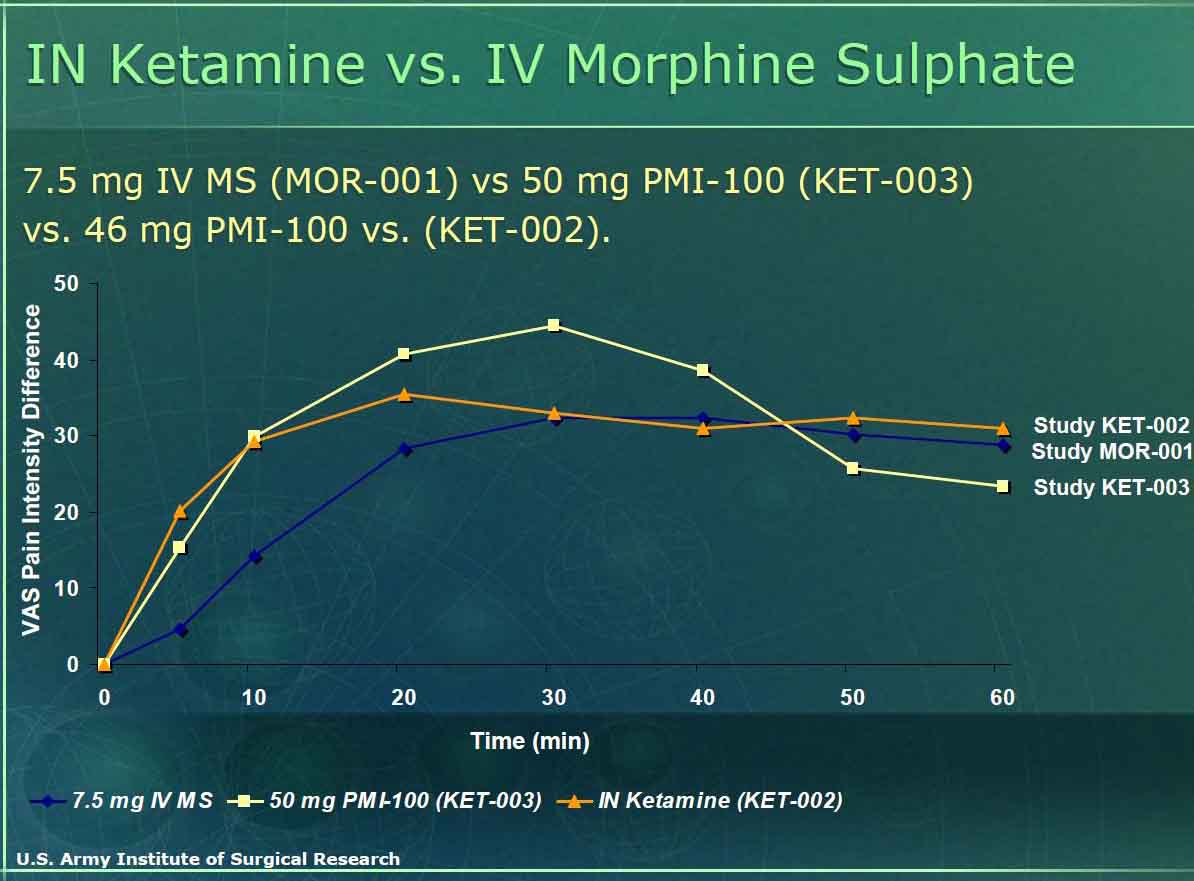
Summary regarding intranasal ketamine:
Intranasal ketamine offers another option for breakthrough pain in situations that are poorly responsive to first line analgesics. Both the patient and the provider can easily administer this medication in a generic form that is readily available and inexpensive. Onset of action is within a few minutes, peaking at 15-30 minutes and wearing off in 2 or more hours. Because it functions via a mechanism and via CNS receptors that are different than opiates it often is helpful in situations where opiates are no longer optimally effective.
Sedation and Acute Seizure control in hospice
Extensive literature exists regarding the use of nasal medications for sedating patients (see sedation section of this web site – click for link). The majority of research on the topic has been directed toward children in pre-operative settings to reduce separation anxiety when taken from their parents, or for pre-procedural sedation to assist with invasive procedures.[36-39] Additional literature exists for anxiolysis in both adults and children prior to radiographic studies or for dental procedures.[40-46] There is even literature in volunteers documenting the reliable absorption of intranasal haloperidol – the authors concluding that this could be a useful alternative to treat acute psychosis in an otherwise uncooperative patient.[47] Finally, intranasal benzodiazepines are well documented to work for the treatment of acute seizures – further confirming their reliable distribution into the central nervous system (Click here for extensive discussion on this topic).[48-50]
At the time of this writing (November 2009) no studies have been published regarding the use of intranasal sedatives or antipsychotics in the setting or agitation or psychosis at the end of life. However, numerous protocols exist suggesting that oral transmucosal benzodiazepines are effective in this situation (see www.palliativedrugs.com). It therefor only makes sense that nasal delivery of lipophilic sedatives and antipsychotics should be effective in this patient population. In fact, based on all comparative trials, nasal absorption tends to outperform oral transmucosal (sublingual and/or buccal) in terms of time of onset, total bioavailability and peak drug levels for opiates and benzodiazepines so the nasal route probably would outperform oral transmucosal for sedation as well. Until this is studied this is purely conjecture based on best available evidence in alternate situations. Perhaps someone who is reading this will conduct a study to confirm or refute this hypothesis.
Home therapy of acute seizures
The data supporting the safety and efficacy of intranasal benzodiazepines for rapid treatment of seizures in the outpatient setting (home, extended care facility, ambulance) is compelling (and extensively reviewed in another section of this web site – click here for intranasal medications for seizures). While none of these studies was conducted in dying patients (except the Ahmad study[50] where 20% of the rural African children in the study died due to underlying infectious diseases) there is no reason to suspect that intranasal benzodiazepines will not be effective in this patient population. However – there is one caveat concerning nasal midazolam in adults – volume of distribution. Midazolam in the current generic formulation of 5 mg/ml is perhaps too dilute for reliable efficacy to stop seizures in adults (based on adult literature it does work, never the less it still requires a high volume for the nasal cavity.) To optimize antiseizure activity it might be a good idea to compound midazolam for a patient with frequent seizures to a concentration of 25 to 50 mg/ml (similar to the concentrations used by Manley for sedation in mentally disabled adults).[43]At these concentrations an adult patient can easily be administered 0.2 mg/kg of nasal midazolam in an appropriate volume for the nose. The compounded drug could them be stored in a multidose bottle delivery system with a pump/atomizer tip that delivers 0.1 ml per spray (2.5 to 5 mg per spray) allowing easy delivery of the appropriate dose and volume for an acute seizure. The slight added cost for compounding the medication in this fashion would probably easily be offset by the savings in reduced ambulance and emergency room visit(s).There is certainly little downside to administering benzodiazepines intranasally for acute seizures. They are shown to be as effective as intravenous benzodiazepines but faster in onset (due to no delays in establishing an IV).[51] They cause minimal sedation and actually reduce need for airway management compared to failure to treat (or treatment with rectal drugs).[49] Finally, because they can be easily administered by family members they will be given much sooner than other treatment options. By improving time to treatment and resolution seizures are less apt to persist and easier to control.
Teaching Materials
Updated slide set Nov 2012:
Intranasal medications for Hospice
Older slide set: Advantages of intranasal medications in the hospice patient
Dose titration protocol for Intranasal fentanyl (or sufentanil) in the hospice patient
Photo guide for method of delivering intranasal medications in the home hospice setting
Intranasal Medications in Hospice - PowerPoint Slides (2.5 MB)
Breakthrough pain lecture by Dr. Portenoy from Beth Isreal Hospital, NY
Review article regarding Instanyl for breakthrough pain: Chaplin 2010
Bibliography (click here for abstracts)
1. Good, P., et al., Intranasal sufentanil for cancer-associated breakthrough pain. Palliat Med, 2009. 23(1): p. 54-8.
2. Carr, D.B., et al., Safety and efficacy of intranasal ketamine for the treatment of breakthrough pain in patients with chronic pain: a randomized, double-blind, placebo-controlled, crossover study. Pain, 2004. 108(1-2): p. 17-27.
3. Rauck, R.L., et al., Efficacy and long-term tolerability of sublingual fentanyl orally disintegrating tablet in the treatment of breakthrough cancer pain. Curr Med Res Opin, 2009.
4. Kress, H.G., et al., Efficacy and tolerability of intranasal fentanyl spray 50 to 200 microg for breakthrough pain in patients with cancer: a phase III, multinational, randomized, double-blind, placebo-controlled, crossover trial with a 10-month, open-label extension treatment period. Clin Ther, 2009. 31(6): p. 1177-91.
5. Watanabe, S., et al., A randomized double-blind crossover comparison of continuous and intermittent subcutaneous administration of opioid for cancer pain. J Palliat Med, 2008. 11(4): p. 570-4.
6. Mercadante, S., et al., Sustained-release oral morphine versus transdermal fentanyl and oral methadone in cancer pain management. Eur J Pain, 2008. 12(8): p. 1040-6.
7. Slatkin, N.E., et al., Fentanyl buccal tablet for relief of breakthrough pain in opioid-tolerant patients with cancer-related chronic pain. J Support Oncol, 2007. 5(7): p. 327-34.
8. Mercadante, S., et al., Transmucosal fentanyl vs intravenous morphine in doses proportional to basal opioid regimen for episodic-breakthrough pain. Br J Cancer, 2007. 96(12): p. 1828-33.
9. Portenoy, R.K., et al., Fentanyl buccal tablet (FBT) for relief of breakthrough pain in opioid-treated patients with chronic low back pain: a randomized, placebo-controlled study. Curr Med Res Opin, 2007. 23(1): p. 223-33.
10. Portenoy, R.K., et al., A randomized, placebo-controlled study of fentanyl buccal tablet for breakthrough pain in opioid-treated patients with cancer. Clin J Pain, 2006. 22(9): p. 805-11.
11. Zeppetella, G. and M.D. Ribeiro, Opioids for the management of breakthrough (episodic) pain in cancer patients. Cochrane Database Syst Rev, 2006(1): p. CD004311.
12. Mercadante, S., et al., A comparison of intranasal fentanyl spray with oral transmucosal fentanyl citrate for the treatment of breakthrough cancer pain: an open-label, randomised, crossover trial. Curr Med Res Opin, 2009. 25(11): p. 2805-15.
13. Farrar, J.T., et al., Oral transmucosal fentanyl citrate: randomized, double-blinded, placebo-controlled trial for treatment of breakthrough pain in cancer patients. J Natl Cancer Inst, 1998. 90(8): p. 611-6.
14. Coluzzi, P.H., et al., Breakthrough cancer pain: a randomized trial comparing oral transmucosal fentanyl citrate (OTFC) and morphine sulfate immediate release (MSIR). Pain, 2001. 91(1-2): p. 123-30.
15. Chien, Y.W., K.S.E. Su, and S.F. Chang, Chapter 1: Anatomy and Physiology of the Nose., in Nasal Systemic Drug Delivery, Y.W. Chien, K.S.E. Su, and S.F. Chang, Editors. 1989, Marcel Dekker Inc,: New York. p. 1-26.
16. Stanley, T.H., Anesthesia for the 21st Century. BUMC Proceedings, 2000. 13: p. 7-10.
17. Schwagmeier, R., et al., Pharmacokinetics of intranasal alfentanil. J Clin Anesth, 1995. 7(2): p. 109-13.
18. Osborn, H. and M. Jefferson, Intranasal alfentanil for severe intractable angina in inoperable coronary artery disease. Palliat Med, 2009.
19. Dale, O., R. Hjortkjaer, and E.D. Kharasch, Nasal administration of opioids for pain management in adults. Acta Anaesthesiol Scand, 2002. 46(7): p. 759-70.
20. Denby, A., Fentanyl preparations for breakthrough cancer pain. London New Drugs Group APC/DTC briefing, 2009. August 2009: p. 1-9.
21. Zeppetella, G., An assessment of the safety, eff icacy, and acceptability of intranasal fentanyl citrate in the management of cancer-related breakthrough pain. A pilot study. J Pain Symptom Manage, 2000. 20(4): p. 253-8.
22. Zeppetella, G., Nebulized and intranasal fentanyl in the management of cancer-related breakthrough pain. Palliat Med, 2000. 14(1): p. 57-8.
23. Sitte, T. and C. Bausewein, Intranasal fentanyl for episodic breathlessness. J Pain Symptom Manage, 2008. 36(6): p. e3-6.
24. Harlos, M., Palliative Care Incident Pain and Incident Dyspnea Protocol. Internet public domain protocol, 2002: p. http://palliative.info/incidentpain.htm.
25. Jackson, K., M. Ashby, and J. Keech, Pilot dose finding study of intranasal sufentanil for breakthrough and incident cancer-associated pain. J Pain Symptom Manage, 2002. 23(6): p. 450-2.
26. Gardner-Nix, J., Oral transmucosal fentanyl and sufentanil for incident pain. J Pain Symptom Manage, 2001. 22(2): p. 627-30.
27. Stoker, D.G., et al., Analgesic efficacy and safety of morphine-chitosan nasal solution in patients with moderate to severe pain following orthopedic surgery. Pain Med, 2008. 9(1): p. 3-12.
28. Kendall, J.M., B.C. Reeves, and V.S. Latter, Multicentre randomised controlled trial of nasal diamorphine for analgesia in children and teenagers with clinical fractures. Bmj, 2001. 322(7281): p. 261-5.
29. Telfer, P.T., et al., Intranasal diamorphine for acute sickle cell pain. Arch Dis Child, 2009.
30. Jackson, K., et al., "Burst" ketamine for refractory cancer pain: an open-label audit of 39 patients. J Pain Symptom Manage, 2001. 22(4): p. 834-42.
31. Mercadante, S., et al., Analgesic effect of intravenous ketamine in cancer patients on morphine therapy: a randomized, controlled, double-blind, crossover, double-dose study. J Pain Symptom Manage, 2000. 20(4): p. 246-52.
32. Knox, D.J., B.J. McLeod, and C.R. Goucke, Acute phantom limb pain controlled by ketamine. Anaesth Intensive Care, 1995. 23(5): p. 620-2.
33. Kulbe, J., The use of ketamine nasal spray for short-term analgesia. Home Healthc Nurse, 1998. 16(6): p. 367-70.
34. Carr, et al., Safety and efficacy of intranasal ketamine in a mixed population with chronic pain. Pain, 2004. 110(3): p. 762-4.
35. Huge, V., et al., Effects of low-dose intranasal (S)-ketamine in patients with neuropathic pain. Eur J Pain, 2009.
36. Lane, R.D. and J.E. Schunk, Atomized intranasal midazolam use for minor procedures in the pediatric emergency department. Pediatr Emerg Care, 2008. 24(5): p. 300-3.
37. Talon, M.D., et al., Intranasal Dexmedetomidine Premedication is Comparable With Midazolam in Burn Children Undergoing Reconstructive Surgery. J Burn Care Res, 2009. 30(4): p. 599-605.
38. Lazol, J.P. and C.G. DeGroff, Minimal sedation second dose strategy with intranasal midazolam in an outpatient pediatric echocardiographic setting. J Am Soc Echocardiogr, 2009. 22(4): p. 383-7.
39. Malinovsky, J.M., et al., Premedication with midazolam in children. Effect of intranasal, rectal and oral routes on plasma midazolam concentrations. Anaesthesia, 1995. 50(4): p. 351-4.
40. Tschirch, F.T., et al., Low-dose intranasal versus oral midazolam for routine body MRI of claustrophobic patients. Eur Radiol, 2007. 17(6): p. 1403-10.
41. Tschirch, F.T., et al., Multicenter trial: comparison of two different formulations and application systems of low-dose nasal midazolam for routine magnetic resonance imaging of claustrophobic patients. J Magn Reson Imaging, 2008. 28(4): p. 866-72.
42. Hollenhorst, J., et al., Using intranasal midazolam spray to prevent claustrophobia induced by MR imaging. AJR Am J Roentgenol, 2001. 176(4): p. 865-8.
43. Manley, M.C., et al., Retrospective audit of the efficacy and safety of the combined intranasal/intravenous midazolam sedation technique for the dental treatment of adults with learning disability. Br Dent J, 2008.
44. Fukuta, O., et al., The sedative effect of intranasal midazolam administration in the dental treatment of patients with mental disabilities. Part 1. The effect of a 0.2 mg/kg dose. J Clin Pediatr Dent, 1993. 17(4): p. 231-7.
45. Fukuta, O., et al., The sedative effects of intranasal midazolam administration in the dental treatment of patients with mental disabilities. Part 2: optimal concentration of intranasal midazolam. J Clin Pediatr Dent, 1994. 18(4): p. 259-65.
46. Kaufman, E., et al., Comparison between intranasal and intravenous midazolam sedation (with or without patient control) in a dental phobia clinic. J Oral Maxillofac Surg, 1994. 52(8): p. 840-3.
47. Miller, J.L., et al., Comparison of intranasal administration of haloperidol with intravenous and intramuscular administration: a pilot pharmacokinetic study. Pharmacotherapy, 2008. 28(7): p. 875-82.
48. Wolfe, et al., Intranasal midazolam therapy for pediatric status epilepticus. Am J Emerg Med, 2006. 24(3): p. 343-6.
49. Holsti, M., et al., Prehospital intranasal midazolam for the treatment of pediatric seizures. Pediatr Emerg Care, 2007. 23(3): p. 148-53.
50. Ahmad, S., et al., Efficacy and safety of intranasal lorazepam versus intramuscular paraldehyde for protracted convulsions in children: an open randomised trial. Lancet, 2006. 367(9522): p. 1591-7.
51. Lahat, E., et al., Comparison of intranasal midazolam with intravenous diazepam for treating febrile seizures in children: prospective randomised study. Bmj, 2000. 321(7253): p. 83-6.
52. Taylor, D.R., Fisher A.N., Smith A. (2009). Pharmacokinetic and local tolerability profiles of three potential fentanyl nasal spray formulations developed for breakthrough cancer pain compared with oral transmucosal fentanyl citrate. J Clinical Oncology 27 (15S) Abstract e20554
53. Kress, H.G., Intranasal fentanyl spray for breakthrough cancer pain: An innovative approach to the unmet needs. European Journal of Pain Supplements, 2010: p. In press.
 Therapeutic
Intranasal Drug Delivery
Therapeutic
Intranasal Drug Delivery